In a nutshell
Prescribed with appropriate screening and monitoring, TCAs are manageable for most non-geriatric patients. The cardiac danger of TCAs is primarily a high-dose phenomenon. At therapeutic doses, TCAs do not appear to meaningfully increase the risk of sudden cardiac death. The three risks that most consistently require active management are overdose lethality, anticholinergic burden in older adults, and wide pharmacokinetic variability driven by CYP2D6 genotype.
This guide covers safety, monitoring, and management; for indications, agent selection, dosing, and titration, see TCA Part 1: Selection and Prescribing.

- Before prescribing:
- Assess suicide and overdose risk: TCAs are the most lethal antidepressant class in overdose [1,2]
- Limit dispensed quantity (e.g., 1-week supply initially)
- Involve family or caregiver
- Choose a less lethal agent if risk is present
- Obtain a baseline ECG[3–5]
- In all patients ≥40 years
- At any age with cardiac history, QT-prolonging co-medications, or electrolyte risk
- If QTc >500 ms or QRS >100 ms: obtain cardiology input before initiation [4–6]
- Check baseline potassium[3,5]
- Hypokalemia potentiates QTc prolongation; correct to ≥3.5 mEq/L before initiating
- Review concomitant medications
- Strong CYP2D6 inhibitors (fluoxetine, paroxetine, bupropion) can raise TCA levels 3–8× [6–8]
- QT-prolonging and serotonergic drugs warrant caution
- In older adults[9]
- Calculate the Anticholinergic Cognitive Burden (ACB) score across all current medications
- If ACB ≥3, strongly prefer nortriptyline or desipramine or a different antidepressant class
- Consider CYP2D6 genotyping if available (non-routine; reserve for suspected extreme metabolizer status, prior unexplained intolerance, or before high-dose escalation) [7]
- Poor metabolizers face up to 8-fold AUC increases
- Ultrarapid metabolizers may never reach therapeutic levels
- Assess suicide and overdose risk: TCAs are the most lethal antidepressant class in overdose [1,2]
- Common side effects:
- Anticholinergic effects (dry mouth, constipation, blurred vision):
- Worst with amitriptyline and doxepin [10]
- If significant, switch class or prefer nortriptyline or desipramine
- Sedation:
- Worst with doxepin and amitriptyline [10]
- Give the full dose at bedtime to convert a side effect into a feature
- Orthostatic hypotension:
- Worst with imipramine; lowest with nortriptyline and desipramine [10–12]
- Bedtime dosing, slow position changes; switch agent if persistent
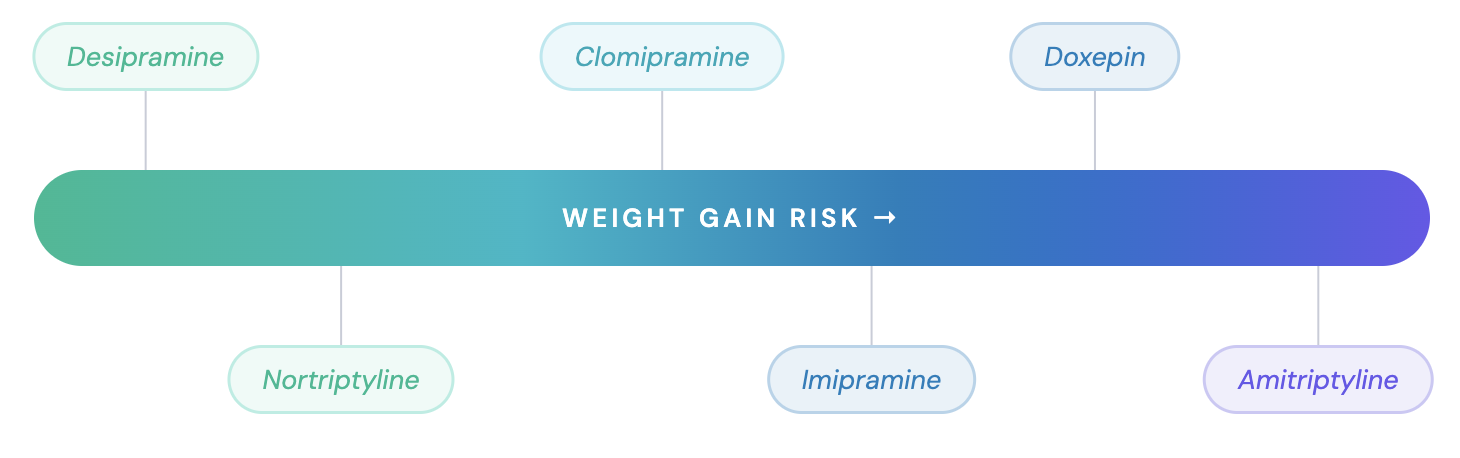
- Weight gain:
- Worst with amitriptyline and doxepin; lowest with nortriptyline and desipramine [10,13]
- When tolerability is the priority, default to nortriptyline or desipramine:
- Lowest anticholinergic burden, lowest orthostatic hypotension, and the best-characterized TDM window in the class [10,11,14]
- Anticholinergic effects (dry mouth, constipation, blurred vision):
- Cardiac risk at therapeutic doses:
- Most TCAs may not meaningfully increase the risk of myocardial infarction or sudden cardiac death when used at normal clinical doses [2]
- Risk is concentrated in overdose, high doses (≥300 mg/day equivalents), and susceptible patients (pre-existing cardiac disease, electrolyte abnormalities, QT-prolonging drugs) [2,15]
- Overdose: the genuine lethal risk:
- TCAs have a narrow therapeutic index
- Lethal effects can occur at roughly 3–5× the usual daily dose, often less than a week’s supply [16,17]
- QRS duration is the primary ECG screening marker of severe TCA toxicity; sodium bicarbonate is the cornerstone of management [18]
- Therapeutic drug monitoring (TDM) at steady state:
- Draw a trough level after ~1–2 weeks at a stable dose (at least 3 weeks for clomipramine), 12 h ± 2 h after the last dose [14]
- For tertiary amines, always request parent compound + active metabolite together (e.g., amitriptyline + nortriptyline; clomipramine + desmethylclomipramine; imipramine + desipramine) [14]
- Repeat TDM after any dose change and after starting/stopping a CYP2D6 inhibitor
Pharmacokinetics and Drug Interactions
Metabolism

- TCAs undergo extensive hepatic first-pass metabolism (~50% average bioavailability) [7]
- Metabolic pathways involve two sequential steps:
- Demethylation (CYP2C19 primary, CYP1A2, CYP3A4): Converts tertiary amines to pharmacologically active secondary amine metabolites [7]
- Amitriptyline → nortriptyline
- Imipramine → desipramine
- Clomipramine → desmethylclomipramine
- Shifts receptor profile from serotonergic (parent) to noradrenergic (metabolite)
- Hydroxylation (CYP2D6 primary):
- Both parent compounds and secondary amine metabolites are hydroxylated, undergo glucuronide conjugation, and are renally excreted [7]
- Demethylation (CYP2C19 primary, CYP1A2, CYP3A4): Converts tertiary amines to pharmacologically active secondary amine metabolites [7]
- At higher doses, dose-to-plasma-level proportionality can break down; small dose increases may produce disproportionate rises in concentration.
- Use therapeutic drug monitoring when titrating toward the upper dose range [7]
- CYP2D6 and CYP2C19 genotyping can guide TCA dosing when results are available [7]
- CYP2D6 poor metabolizers (genetic or drug-induced via inhibition) can show up to 8-fold higher AUC of the TCA compared with normal metabolizers [6,19]
Drug Interactions
Pharmacokinetic Interactions
- TCA levels increased by:
- CYP2D6 inhibitors
- Can raise TCA plasma concentrations 3- to 8-fold, potentially precipitating toxicity in previously stable patients [6–8]
- Strong inhibitors: fluoxetine, paroxetine, bupropion, quinidine
- Quinidine specifically increases nortriptyline AUC and half-life [20]
- Fluoxetine washout: given the long half-life of fluoxetine and norfluoxetine, at least 5 weeks should elapse after discontinuation before initiating a TCA [6,19]
- Management: avoid co-administration when possible; if unavoidable, reduce TCA dose by ≥50% with close clinical monitoring and TDM
- CYP1A2 inhibitors
- Raise tertiary amine TCA levels through a pathway distinct from CYP2D6
- Fluvoxamine is a strong CYP1A2 inhibitor [8]
- Agent-specific interactions:
- Topiramate may cause large increases in amitriptyline concentration; adjust amitriptyline dose based on clinical response (relevant when both are used for migraine) [6]
- Haloperidol increases clomipramine plasma concentrations [19]
- CYP2D6 inhibitors
- TCA levels decreased by:
- CYP1A2 inducers:
- Smoking, carbamazepine; reduce tertiary amine TCA levels [8]
- Clinical pearl: smoking cessation (hospitalization, quit attempt) removes CYP1A2 induction within days and can raise TCA levels into the toxic range.
- Monitor TDM and consider proactive dose reduction
- CYP1A2 inducers:
- Effects of TCAs on other drugs:
- Warfarin:
- TCAs may increase warfarin plasma concentrations (likely via protein-binding displacement); monitor INR when initiating, stopping, or titrating a TCA [19]
- Guanethidine, clonidine, and similar antihypertensives:
- TCAs block their antihypertensive action [6,19,20]
- Warfarin:
Pharmacodynamic Interactions
- Monoamine oxidase inhibitors (MAOIs)
- Contraindicated. Risk of serotonin syndrome and hypertensive crisis
- A 14-day washout is required when switching between TCAs and MAOIs [6,19,20]
- Linezolid and intravenous methylene blue have MAOI-like activity and should be avoided (access the full Serotonin Syndrome Guide)
- Serotonergic drugs (SSRIs, SNRIs, tramadol, triptans, St. John’s wort)
- Increased risk of serotonin syndrome
- Clomipramine and imipramine carry greater risk due to higher SERT affinity [21]
- Tramadol + clomipramine is a particularly high-risk combination (combines serotonergic load with seizure-threshold lowering) and should be avoided [19,21]
- QT-prolonging drugs
- Additive QTc prolongation and arrhythmia risk
- Avoid or use with careful ECG monitoring alongside:
- Class 1A (quinidine, procainamide, disopyramide)
- Class 3 antiarrhythmics (amiodarone, sotalol)
- Methadone
- Certain antipsychotics (haloperidol, thioridazine, ziprasidone)
- Macrolides; fluoroquinolones
- Ondansetron
- Anticholinergic agents
- Increases risk of constipation, urinary retention, blurred vision, cognitive impairment, and delirium
- Hyperpyrexia has been reported with TCA + anticholinergic or TCA + neuroleptic combinations, particularly in hot weather [6]
- Sympathomimetics
- TCAs potentiate the pressor effect of direct-acting sympathomimetics (norepinephrine, epinephrine) and blunt the response to indirect-acting agents (phenethylamine, ephedrine) [6,20]
Contraindications
- Concurrent MAOI use (or within 14 days of discontinuation) [6,19,20]
- Acute recovery phase after myocardial infarction [6,19,20]
- Narrow-angle glaucoma or anatomically narrow anterior-chamber angles without a patent iridectomy [6,20]
- Brugada type 1 pattern [20]
- Avoid TCAs or obtain cardiology input in patients with significant conduction disease (eg, bundle branch block or 2°/3° AV block), given the risk of worsening conduction abnormalities [22]
Side effects
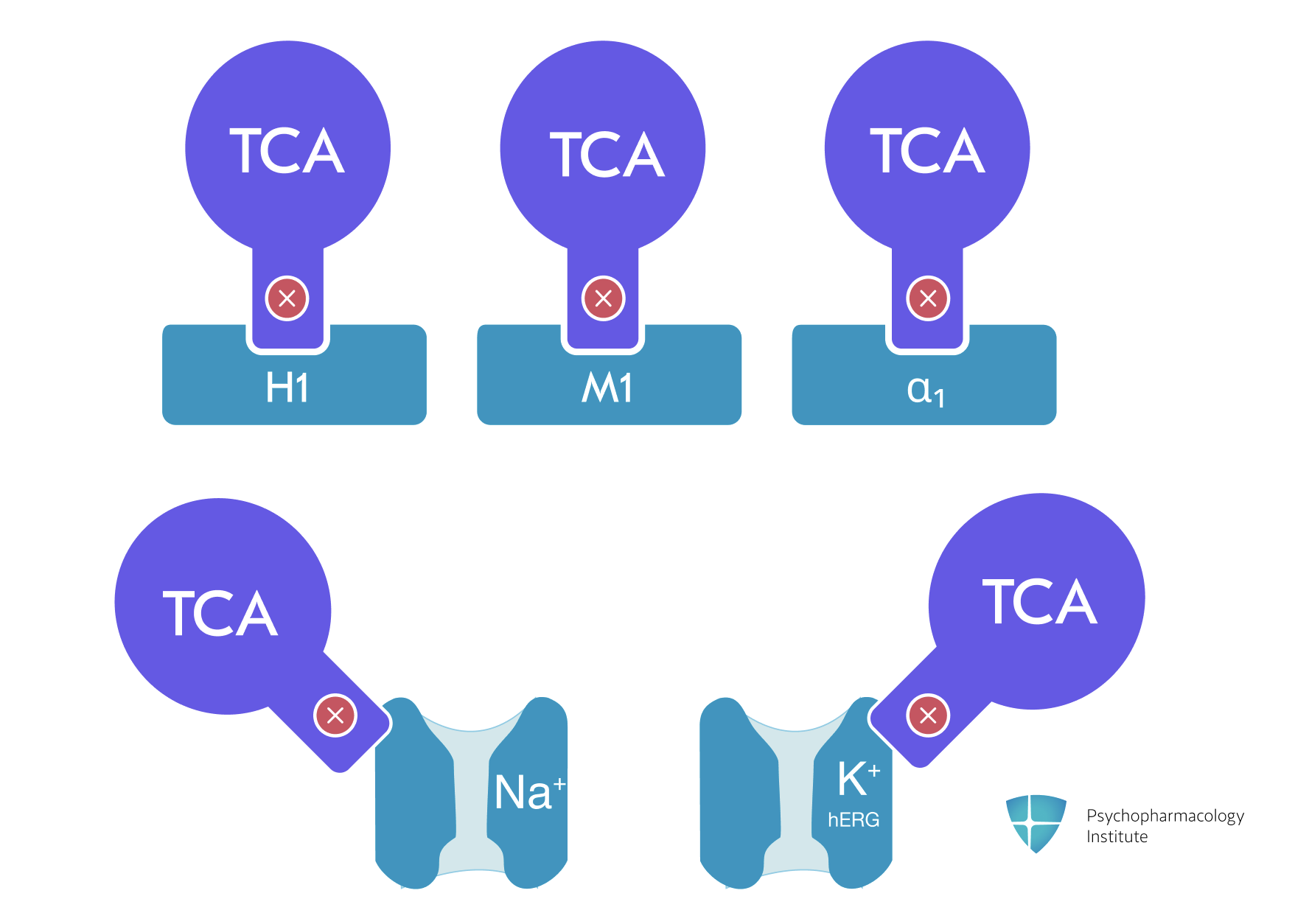
- TCAs share a broader side-effect profile than SSRIs/SNRIs due to their multi-receptor pharmacology:
- H1 antagonism (sedation, weight gain)
- Muscarinic blockade (anticholinergic effects)
- Alpha-1 antagonism (orthostatic hypotension)
- Sodium channel blockade (cardiac risk)
- The severity and pattern of side effects vary predictably with the tertiary vs secondary amine distinction [10,16]
- Tertiary amines (amitriptyline, clomipramine, doxepin, imipramine, trimipramine) cause more anticholinergic, sedative, and cardiovascular effects
- Secondary amines (nortriptyline, desipramine, protriptyline) are generally better tolerated.
Anticholinergic effects
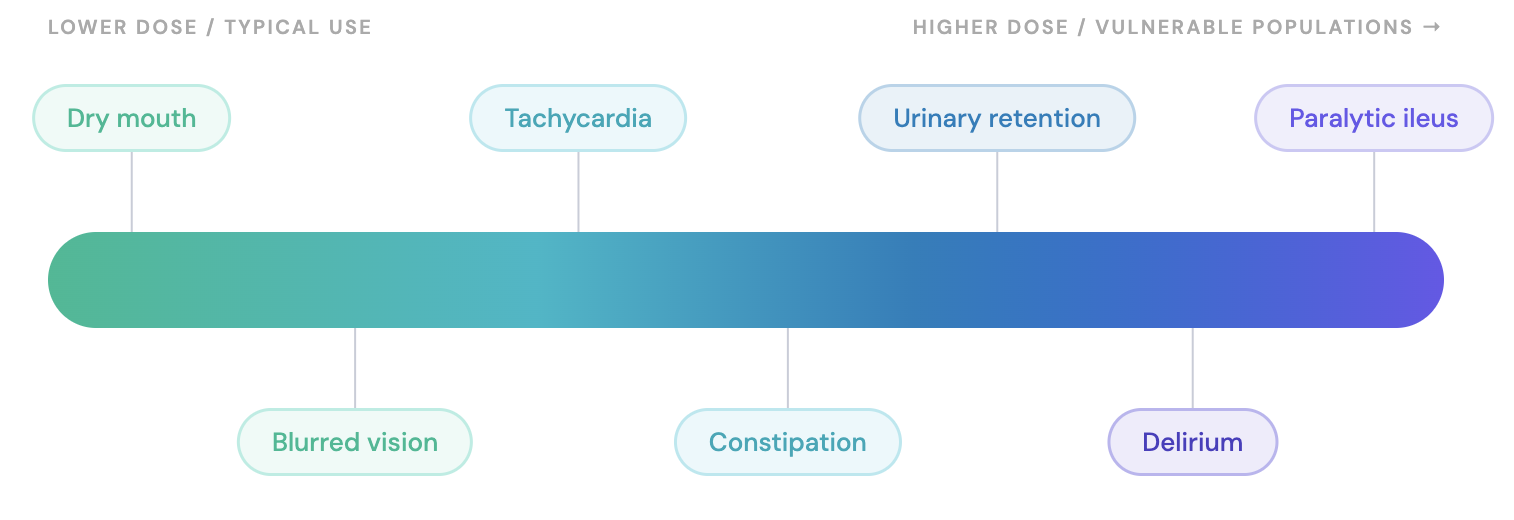
- Spectrum ranges in a dose-dependent manner from dry mouth and constipation to urinary retention and blurred vision [6,19,20]
- Can progress to tachycardia, paralytic ileus, or delirium at higher doses or in vulnerable populations (elderly, polypharmacy with other anticholinergics)
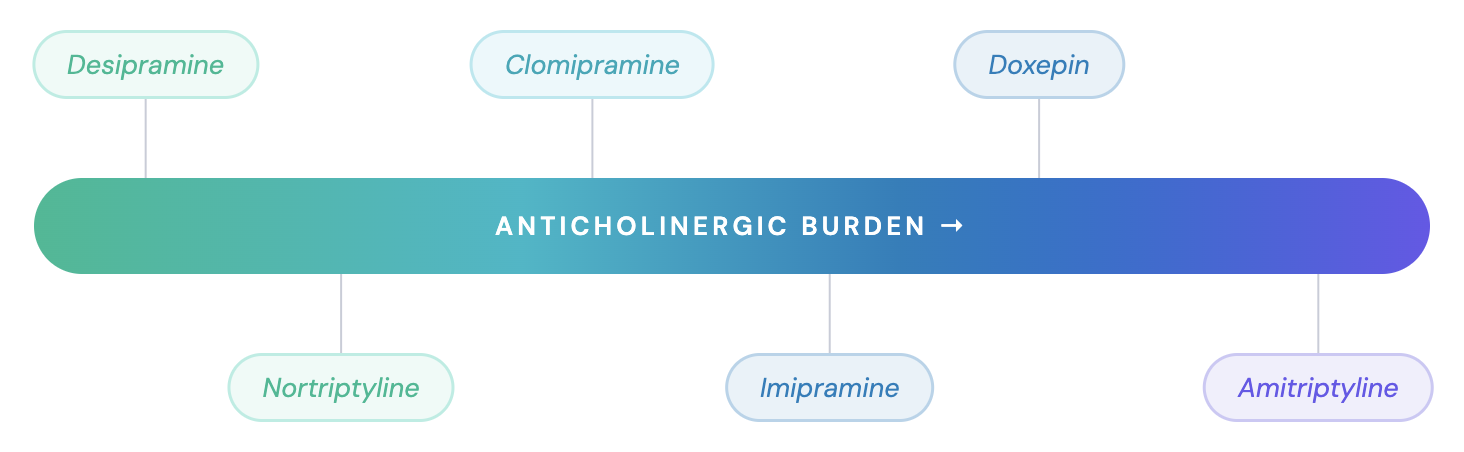
- Comparative anticholinergic burden by agent, lowest to highest:
- Desipramine < Nortriptyline < Imipramine < Clomipramine < Doxepin < Amitriptyline [10]
- Cumulative anticholinergic exposure has been linked to an increased risk of incident dementia [23,24]
- The American Geriatrics Society Beers Criteria list tertiary amine TCAs as potentially inappropriate in older adults (≥65 years) [9]
- Management: (access full guide: Gastrointestinal Side Effects of Antidepressants: Mechanisms, Comparison and Management Strategies)
Cardiovascular effects
- TCAs inhibit voltage-sensitive sodium channels (causing QRS prolongation) and hERG potassium channels (causing QTc prolongation) [2]
- This dual ion-channel activity is the mechanistic basis for both therapeutic-dose conduction changes and overdose cardiotoxicity
- Orthostatic hypotension
- The most common cardiovascular effect and one of the primary reasons for treatment discontinuation [12]
- Falls in elderly, reflex tachycardia, syncope
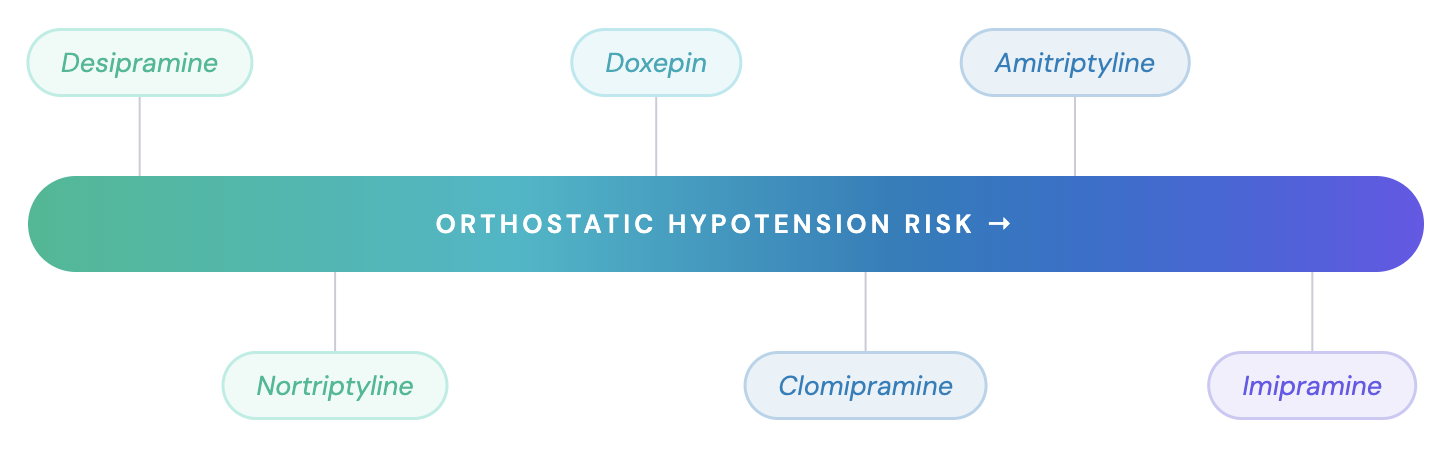
- Agent ranking, lowest to highest risk:
Desipramine ≈ Nortriptyline < Doxepin < Clomipramine < Amitriptyline < Imipramine [10–12] - Management:
- Slow position changes; adequate hydration; elastic stockings
- Give full dose at bedtime
- If persistent despite these measures: switch to nortriptyline or desipramine (lowest α1 blockade in class) [11]
- Cardiac conduction changes
- QRS widening, PR prolongation, and QTc prolongation [2,6,20]
- Risk is greater at toxic doses but can also occur at therapeutic levels in susceptible patients
- All TCAs should generally be avoided, or used only with cardiology input, in patients with pre-existing bundle branch block or advanced AV block [6,19,20,22,25]
- High-dose TCA use (≥300 mg/day amitriptyline or equivalent) has been associated with increased risk of sudden cardiac death, even in patients without known cardiac disease [15]
- For ECG screening thresholds, contraindication criteria, and ongoing cardiac monitoring, see the Monitoring Considerations section
- QRS widening, PR prolongation, and QTc prolongation [2,6,20]
- Sinus tachycardia
- Usually benign and does not require treatment
- Brugada syndrome unmasking
- TCAs with sodium channel-blocking properties can unmask Brugada syndrome in genetically predisposed individuals [20]
- Nortriptyline’s FDA label specifically flags Brugada unmasking as a postmarketing finding [20]
- Clinical perspective: therapeutic-dose cardiac risk in context
- At normal clinical doses, most TCAs may not increase the risk of sudden cardiac death, according to a 2024 systematic review of 14 observational studies [2]
- In-use cardiotoxicity signals were still noted for amitriptyline, nortriptyline, and lofepramine [2]
- This does not eliminate the need for ECG screening: cardiac risk is dose-dependent [2,15]
Other common side effects
- Sedation/drowsiness (H1 blockade)
- Most prominent with doxepin, amitriptyline, imipramine; uncommon with desipramine (see comparative receptor affinity table in the Pharmacodynamics section of TCA Part 1 guide) [10]
- Management:
- Give the full dose at bedtime
- Consider switching to a less sedating agent if problematic
- Nausea, dyspepsia
- More common with clomipramine
- Dividing doses with meals during titration reduces symptoms [19]
- Tremor:
- Common; may respond to dose reduction [6,20]
- Diaphoresis
- Appears to be related to noradrenergic effects
- May be more prominent with secondary amines [1]
- Weight gain and metabolic effects (access the full guide Antidepressants and Metabolic Disturbances)
- H1 antagonism drives increased appetite and weight gain [10]
- Sexual dysfunction
- Decreased libido, erectile dysfunction, ejaculatory dysfunction reported with all TCAs [6,19,20]
- Clomipramine is associated with anorgasmia, possibly due to its potent SERT inhibition [26]
Severe side effects
- Overdose toxicity:
- TCAs are the antidepressant class with the highest morbidity and mortality in overdose [1,2]
- Seizure threshold lowering
- All TCAs lower seizure threshold; risk is dose- and serum-level-dependent [2]
- Clomipramine carries the highest seizure risk in the class; this is a primary reason the FDA caps the recommended dose at 250 mg/day [19]
- Hyponatremia/SIADH
- Clinically significant hyponatremia reported with TCAs, particularly clomipramine and amitriptyline [6,19]
- Monitor sodium in at-risk patients (elderly, concurrent diuretics)
- Serotonin syndrome (access the full Serotonin Syndrome Guide)
- Risk with serotonergic combinations (MAOIs, SSRIs, tramadol)
- Clomipramine carries the highest risk among TCAs due to its potent serotonin reuptake inhibition
- MAOIs are contraindicated with TCAs; a 14-day washout is mandatory when switching between these classes
- Activation of mania/hypomania
- TCAs carry a higher mania switch rate than other antidepressants in bipolar depression [27,28]
- Avoid in bipolar disorder; if an antidepressant is required, bupropion may confer lower switch risk
- Discontinuation symptoms
- Nausea, headache, malaise, irritability, restlessness, and sleep disturbance may occur with abrupt cessation [6,19,20]
- Rare instances of mania or hypomania reported within 2–7 days following abrupt cessation of chronic TCA therapy [6]
- Gradual taper recommended; reduce dose by 25–50% every 1–2 weeks
- Suicidality
- All antidepressants carry an FDA boxed warning for suicidality; exercise special caution given the lethality of TCAs in overdose [6,19,20]
- Liver injury (clomipramine)[19]
- Clomipramine can cause abnormal hepatic function and hepatitis as infrequent adverse events; rare severe liver injury, some fatal, reported in foreign postmarketing experience [19]
- Monitor LFTs (AST, ALT) periodically in patients with known liver disease
- Transaminase elevations >3× ULN are flagged as of potential clinical importance in the label; consider dose reduction or discontinuation and exclude other causes
Use in special populations
Pregnancy
- No confirmed teratogenic risk for most TCAs as a class in humans, though controlled data are limited [29–31]
- General recommendations:
- If a TCA is needed during pregnancy, prefer nortriptyline [29,32–34]
- It has the most reassuring observational data across registries and cohort studies
- Its pharmacokinetics during pregnancy are relatively well characterized within the class
- Monitor serum levels, as TCA concentrations tend to decline during pregnancy.
- Consider neonatal monitoring for withdrawal or adaptation symptoms (irritability, jitteriness, respiratory distress) if used near delivery
- If a TCA is needed during pregnancy, prefer nortriptyline [29,32–34]
| TCA | Birth defect risk | Key considerations |
|---|---|---|
| Nortriptyline | No signals | Preferred. Plasma levels decline during pregnancy; monitor. |
| Amitriptyline | No consistent signal | Plasma levels may decline during pregnancy. |
| Clomipramine | Cardiac malformations (VSD/ASD) in Swedish registry; no causal link | Neonatal complications near delivery; neonatal half-life ≈42 h. |
| Imipramine | No signal | Dose increases may be needed. |
References: Heinonen et al, 1977; Brunel et al, 1994; Huybrechts et al, 2014; Scherf-Clavel et al, 2025; Simon et al, 2002; Leutritz et al, 2023; Källén and Otterblad Olausson, 2006; Ostergaard and Pedersen, 1982; ter Horst et al, 2012; McElhatton et al, 1996; Ericson et al, 1999; Wisner et al, 1993; Altshuler and Hendrick, 1996; Schoretsanitis et al, 2020.
- Neurodevelopmental outcomes:
- Neurobehavioral testing of 80 children exposed in utero to TCAs (including amitriptyline, imipramine, nortriptyline, and clomipramine) showed no differences in IQ or behavior compared to fluoxetine-exposed or unexposed children at ages 16–86 months [35]
- A follow-up study confirmed no adverse effect of TCA exposure on cognitive development
- Duration of maternal depression and number of postpartum episodes negatively impacted IQ and language [36]
Breastfeeding
| TCA | Safety during lactation | Key considerations |
|---|---|---|
| Nortriptyline | Preferred during lactation | Infant concentrations <10% of maternal; no adverse effects reported. |
| Amitriptyline | Probably safe | Case report of sedation and poor feeding in a 15-day-old at maternal 10 mg/day. |
| Clomipramine | Acceptable with caution | Small amounts in breast milk. |
| Imipramine | Probably safe | Small amounts, with desipramine metabolite, in milk. |
References: Uguz, 2021; Sriraman et al, 2015; Weissman et al, 2004; Gentile, 2014; Davanzo et al, 2011; Wisner and Perel, 1991; Wisner et al, 1997; Breyer-Pfaff et al, 1995; Uguz, 2017; LactMed, 2006; Yoshida et al, 1997; Schimmell et al, 1991; Sovner and Orsulak, 1979; Erickson et al, 1979.
Hepatic impairment
- No specific dosing adjustments are provided in FDA labels for amitriptyline, nortriptyline, or clomipramine at antidepressant doses [6,19,20]
- Exception: For doxepin, consider initiating at 3 mg in patients with hepatic impairment [37]
- Clinical approach:
- Use lower starting doses and titrate slowly
- Monitor serum levels when feasible, as clearance may be reduced
- In older adults, hepatic clearance can be reduced by up to 30%, mainly due to reduced hepatic blood flow [14]
Renal impairment
- No specific dosing adjustments are provided in FDA labels for amitriptyline, nortriptyline, or clomipramine at antidepressant doses [6,19,20]
- Dose reduction is generally not required, given the predominantly hepatic metabolism of TCAs
- However, active metabolites (e.g., 10-hydroxynortriptyline, desipramine) are renally eliminated; accumulation may occur in severe renal impairment
- Caution is advised in severe renal impairment: use lower starting doses and consider monitoring serum levels
Older adults
- TCAs are listed as potentially inappropriate medications in older adults in the 2023 American Geriatrics Society Beers Criteria, primarily due to high anticholinergic burden and risk of falls [9]
- All TCA labels recommend lower starting dosages in older adults [6,19,20]
- If a TCA is needed, prefer nortriptyline or desipramine (least anticholinergic and least orthostatic hypotension among commonly used TCAs) [10,11]
- Nortriptyline oral solution allows fine dose titration
Cardiac disease
- TCAs should be avoided in patients with a history of or at high risk for coronary heart disease [3,22]
Monitoring Considerations
- A practical monitoring framework may help in the prescription process
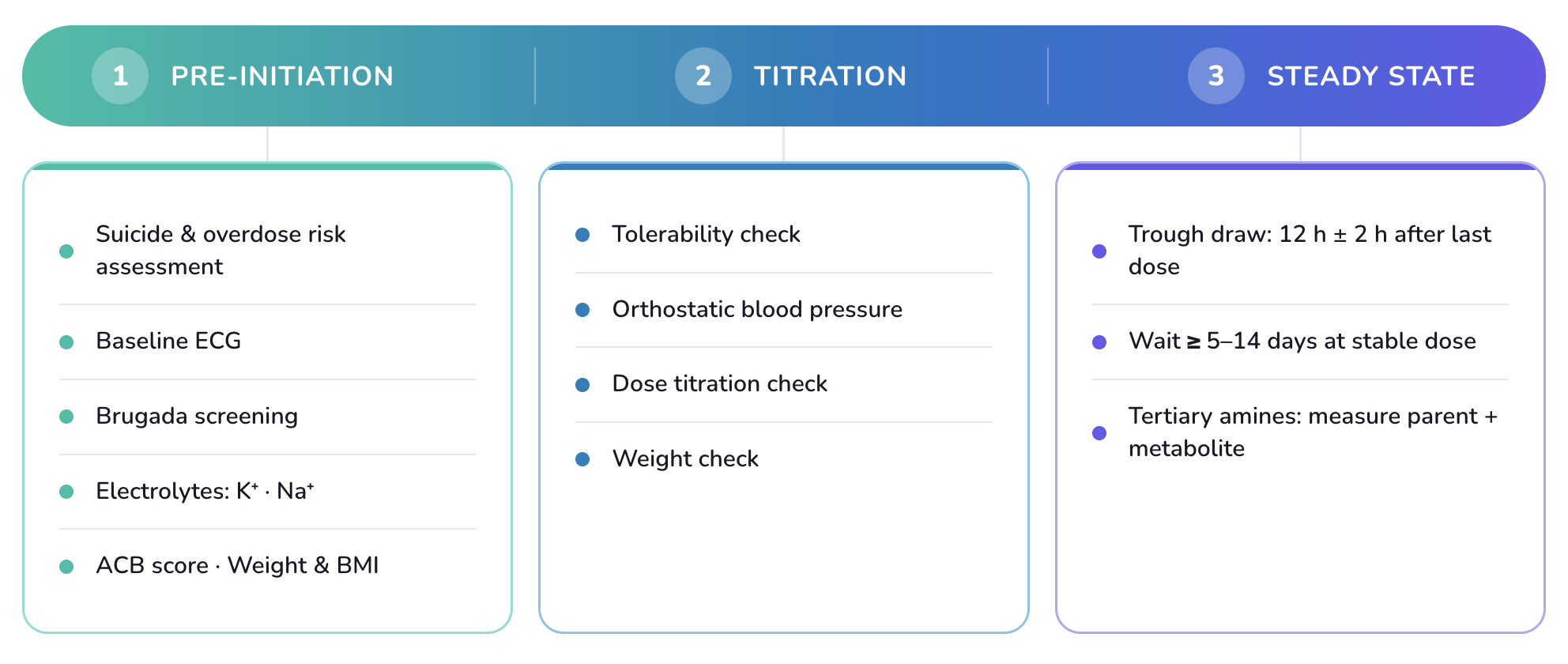
Pre-initiation
- Suicide and overdose risk assessment
- If risk is present: limit dispensed quantity (e.g., 1-week supply initially); involve family or caregiver; document clearly; consider a less lethal alternative
- Medication review
- Screen specifically for concomitant medications that can meaningfully raise TCA exposure or generate pharmacodynamic risk before initiating
- Baseline ECG[3–6]
- Recommended for patients ≥40 years
- Obtain at any age if:
- Cardiac history
- Concurrent QT-prolonging drugs
- Electrolyte disturbance risk (diuretics, vomiting)
- Family history of sudden cardiac death
- Measure QRS, QTc, PR interval; look for bundle branch block and Brugada pattern
- Thresholds:
- QTc >500 ms: do not initiate without cardiology clearance [4,5]; this threshold is associated with substantially increased risk of torsade de pointes
- Baseline QRS prolongation (eg, >100 ms): evaluate for underlying conduction disease and obtain cardiology input before initiation
- Bundle branch block or 2°/3° heart block:
- Avoid TCAs or obtain cardiology input in patients with significant conduction disease [22,25]
- Brugada screening
- Screen by history: personal or family history of unexplained syncope, ventricular arrhythmia, or sudden cardiac death in a first-degree relative <45 years [20]
- If positive, refer to cardiology before initiating a TCA
- Nortriptyline’s FDA label specifically flags Brugada unmasking as a postmarketing finding; the risk extends to all TCAs with sodium channel blockade [20]
- Baseline electrolytes
- Serum potassium: hypokalemia potentiates QTc prolongation — correct to ≥3.5 mEq/L before initiating [3,5]
- Serum sodium: obtain in older adults or patients on diuretics (establishes baseline for SIADH monitoring) [6]
- Anticholinergic Cognitive Burden (ACB) assessment in older adults[9]
- Calculate cumulative ACB score across current medications
- If ACB is already ≥3 before adding a TCA: strongly prefer nortriptyline or desipramine (lower anticholinergic burden) over tertiary amines, or consider a different antidepressant class entirely
- Document baseline ACB score for longitudinal tracking
- Weight and BMI
- Record baseline weight and BMI for comparison at subsequent visits
- TCAs (particularly amitriptyline and doxepin) carry high risk for weight gain (see Antidepressants and Metabolic Disturbances)
First 2 weeks: Titration
- Tolerability check[6,20]
- Phone or visit at day 7–10 after initiation; in older adults, check at day 3–5
- Dose-limiting side effects peak in the first 2 weeks; early identification prevents dropout
- Orthostatic blood pressure
- Especially important in older adults and with tertiary amines (amitriptyline, imipramine, clomipramine), where α1-adrenergic blockade is most pronounced [12,38]
- Measure supine and standing after 1–3 minutes
- Drop ≥20 mmHg systolic or ≥10 mmHg diastolic = orthostatic hypotension
- If symptomatic:
- Reinforce slow position changes, ensure hydration
- Reduce dose or switch to nortriptyline or desipramine (lower α1 blockade)
- Review concurrent antihypertensives
- ECG[4]
- Consider repeating after reaching target dose if:
- Baseline ECG was borderline (QTc 450–500 ms)
- Patient is ≥40 years or has cardiac risk factors
- Dose is approaching upper range (e.g., amitriptyline ≥150 mg/day)
- Action threshold: Dose reduction or cardiology consultation if QTc rise >60 ms from baseline or absolute QTc >500 ms [3,5]
- Consider repeating after reaching target dose if:
Steady state: Therapeutic drug monitoring and interpretation
When and what to measure
- When to draw: after ≥5 half-lives at a stable dose [14]
- Most TCAs: approximately 1–2 weeks at stable dose
- Clomipramine: at least 3 weeks at stable dose (desmethylclomipramine has a longer half-life)
- Timing: trough level, 12 hours (±2 h) after last dose [14]
- What to measure:
- For tertiary amines, always request parent compound + active metabolite together (e.g., amitriptyline + nortriptyline; clomipramine + desmethylclomipramine; imipramine + desipramine) [14]
Therapeutic Reference Ranges
| TCA (measured moiety) | Therapeutic range | Alert level | TDM level | Notes |
|---|---|---|---|---|
| Amitriptyline + nortriptyline | 80–200 ng/mL | 300 ng/mL | 1 (strongly recommended) | Limited data on concentration-response relationship |
| Clomipramine + desmethylclomipramine | 230–450 ng/mL | 450 ng/mL | 1 (strongly recommended) | Parent = serotonergic; metabolite = noradrenergic |
| Nortriptyline | 70–170 ng/mL | 300 ng/mL | 1 (strongly recommended) | Inverted U-shaped: efficacy may decline above upper range |
| Imipramine + desipramine | 175–300 ng/mL | 300 ng/mL | 1 (strongly recommended) | Linear: response more likely at >200 ng/mL |
| Desipramine | 100–300 ng/mL | 300 ng/mL | 2 (recommended) | Linear: response more likely at >125 ng/mL |
| Doxepin + N-desmethyldoxepin | 50–150 ng/mL | 300 ng/mL | 2 (recommended) | Insufficient data for response-concentration modeling |
Reference: Hiemke C, Bergemann N, Clement HW, et al. Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology: Update 2017. Pharmacopsychiatry. 2018;51(1-02):9–62.
Interpreting the results
| Result | Action |
|---|---|
| Below range, adherent | Consider dose increase; if on a CYP2D6 inducer (eg, carbamazepine) or if the patient smokes, expect higher-than-usual doses; consider CYP2D6 ultrarapid metabolizer status. |
| Within range, non-responsive | Reassess diagnosis; consider switching agent or augmentation strategies. |
| Within range, responding | Continue current dose; document level for future reference. |
| Above range | Reduce dose; assess for subclinical toxicity (eg, tremor, tachycardia, QTc change); consider ECG. |
| Markedly supratherapeutic with toxicity signs | Reduce or hold immediately; obtain ECG; hold until the level normalizes; consider CYP2D6 poor metabolizer status or a drug interaction. |
References: Hiemke et al, 2018; Hicks et al, 2017.
Dose changes and drug interaction triggers
- Repeat TDM after any dose change[14]
- Draw new trough level 1–2 weeks after dose change (at least 3 weeks for clomipramine)
- Clomipramine has nonlinear pharmacokinetics above ~150 mg/day [19]
- Dose increases in this range do not produce proportional plasma level increases
- TDM is essential to avoid unexpected accumulation
- Repeat TDM after CYP2D6/CYP2C19 interaction changes[7,14]
- Draw within 2 weeks of adding or removing a CYP2D6 inhibitor or inducer (within 3 weeks for clomipramine)
- Critical scenarios:
- Starting fluoxetine, paroxetine, or bupropion in a patient already on a TCA
- Preemptively reduce TCA dose by ≥50%
- Confirm with TDM [7,14]
- Stopping a CYP2D6 inhibitor
- TCA level will fall as inhibition is removed
- May need dose increase to maintain therapeutic range
- Smoking cessation removes CYP1A2 induction [14]
- Tertiary amine levels (amitriptyline, clomipramine, imipramine) can rise significantly; this is a commonly overlooked interaction
- Consider drawing TDM within 1–2 weeks of quitting
- Starting carbamazepine[14]
- CYP1A2/CYP3A4 induction lowers TCA levels
- Anticipate need for dose increase; confirm with TDM
- Starting fluoxetine, paroxetine, or bupropion in a patient already on a TCA
- Repeat ECG after dose change or new QT interaction
- Repeat ECG if dose increases to upper range or a new QT-prolonging medication is added
- Action threshold remains: QTc rise >60 ms from baseline or absolute QTc >500 ms [5]
Discontinuation
- Taper schedule
- Reduce dose by 25–50% every 1–2 weeks; avoid abrupt cessation [6,19,20]
- Discontinuation symptoms: nausea, headache, malaise, irritability, restlessness, sleep disturbance
- These symptoms typically onset within days of dose reduction and resolve within 1–2 weeks, even without dose restoration
- Mania or hypomania has been described rarely within 2–7 days of abrupt cessation [6]
- Monitor for mood elevation during taper
Pharmacogenomic testing
- Routine preemptive CYP2D6/CYP2C19 genotyping is not currently recommended before initiating a TCA
- CPIC provides dosing guidance if results are available rather than recommending that testing be obtained [7]
- Targeted testing is reasonable in specific scenarios:
- Prior unexplained intolerance, adverse reaction, or non-response to a TCA or another CYP2D6/CYP2C19 substrate
- Before escalating to high-dose TCA treatment (especially tertiary amines)
- TDM showing unexpectedly high or low concentrations despite adherence [14]
- Complex polypharmacy with CYP2D6/CYP2C19 inhibitors or inducers where phenoconversion complicates interpretation [7]
- If genotyping is unavailable: apply clinical phenotyping clues (prior unusual drug sensitivities, CYP2D6 inhibitor exposure history) and rely on TDM
CYP2D6-guided dosing
| Phenotype | Implication | Recommendation | Strength (amitriptyline, nortriptyline) | Strength (other TCAs)* |
|---|---|---|---|---|
| Ultrarapid metabolizer | Increased metabolism; lower plasma concentrations; potential lack of efficacy | Avoid TCA; consider an alternative not metabolized by CYP2D6; if used, guide dose with TDM | Strong | Optional |
| Normal metabolizer | Normal metabolism | Initiate at recommended starting dose | Strong | Strong |
| Intermediate metabolizer | Reduced metabolism; higher plasma concentrations; higher side-effect risk | Consider a 25% reduction of recommended starting dose; guide with TDM | Moderate | Optional |
| Poor metabolizer | Greatly reduced metabolism; substantially higher plasma concentrations (up to 8-fold AUC increase); higher side-effect risk | Avoid TCA; if used, reduce starting dose by 50% with TDM | Strong | Optional |
*Other CYP2D6-metabolized TCAs: clomipramine, desipramine, doxepin, imipramine, trimipramine. Fewer supporting data than for amitriptyline and nortriptyline. Reference: Hicks et al, 2017.
CYP2C19-guided dosing (tertiary amines only)
| Phenotype | Implication | Recommendation | Strength (amitriptyline) | Strength (other tertiary amines)* |
|---|---|---|---|---|
| Ultrarapid or rapid metabolizer | Increased demethylation; higher metabolite-to-parent ratio; potential sub-optimal response | Avoid tertiary amine; consider a drug not metabolized by CYP2C19 (e.g., nortriptyline, desipramine); if used, guide with TDM | Optional | Optional |
| Normal metabolizer | Normal metabolism | Initiate at recommended starting dose | Strong | Strong |
| Intermediate metabolizer | Modestly decreased demethylation | Initiate at recommended starting dose | Strong | Optional |
| Poor metabolizer | Greatly decreased demethylation; higher parent-compound concentrations; higher side-effect risk | Avoid tertiary amine; if used, reduce starting dose by 50% with TDM; or switch to a secondary amine | Moderate | Optional |
*Other CYP2C19-metabolized tertiary-amine TCAs: clomipramine, doxepin, imipramine, trimipramine. Fewer supporting data than for amitriptyline. Reference: Hicks et al, 2017
Overdose Recognition and Management
- TCAs have a narrow therapeutic index: lethal effects can occur at roughly 3–5× the usual daily dose, often less than a week’s supply [16,17]
- Some patients with initially trivial findings can deteriorate rapidly [17]
- ECG is the key monitoring tool
- QRS duration is the primary toxicity marker [18]
- QRS >100 ms: early warning; predictive of seizures and associated with a higher risk of convulsions or arrhythmias
- QRS >160 ms: predictive of ventricular arrhythmias including ventricular tachycardia
- Rightward shift of the terminal QRS is a distinctive sodium-channel-blockade signature, combined with prolonged QT and sinus tachycardia [6]
- A dominant terminal R wave in lead aVR ≥3 mm has been prospectively validated as an independent predictor of seizures and ventricular arrhythmias [39]
- Brugada-type ECG pattern has been reported in approximately 15–17% of TCA overdoses as a postmarketing/case-series finding [20,40]
- The pattern typically resolves after sodium bicarbonate administration [40]
- A normal initial ECG does not exclude serious toxicity, particularly early after ingestion when absorption is incomplete [17]
- QRS duration is the primary toxicity marker [18]
- Plasma drug level monitoring should not guide management
- Levels are often unavailable in real time and do not reliably correlate with toxicity severity at overdose concentrations [17]
Management
- ECG and cardiac monitoring
- Serum alkalinization with sodium bicarbonate [41]
- Gastrointestinal decontamination (activated charcoal within ~2 h of ingestion, airway protected) [42]
- Seizure management
- Benzodiazepines are first-line [17]
- Avoid phenytoin (may worsen sodium channel blockade); barbiturates carry respiratory depression risk [43]
- Contraindicated interventions
- Class IA antiarrhythmics (quinidine, procainamide, disopyramide): worsen sodium channel blockade [6,20]
- Class IC antiarrhythmics (flecainide, propafenone): contraindicated for the same reason [43]
- Flumazenil: may precipitate seizures, especially with mixed overdose involving benzodiazepines [16]
- Physostigmine: despite apparent anticholinergic toxidrome, has been associated with asystole and worsening cardiac toxicity in TCA overdose and should not be used [44]
References
1. Nelson, J. C., & Spyker, D. A. (2017). Morbidity and Mortality Associated With Medications Used in the Treatment of Depression: An Analysis of Cases Reported to U.S. Poison Control Centers, 2000-2014. The American Journal of Psychiatry, 174(5), 438–450. https://doi.org/10.1176/appi.ajp.2016.16050523
2. Taylor, D., Poulou, S., & Clark, I. (2024). The cardiovascular safety of tricyclic antidepressants in overdose and in clinical use. Therapeutic Advances in Psychopharmacology, 14, 20451253241243297. https://doi.org/10.1177/20451253241243297
3. Fanoe, S., Kristensen, D., Fink-Jensen, A., Jensen, H. K., Toft, E., Nielsen, J., Videbech, P., Pehrson, S., & Bundgaard, H. (2014). Risk of arrhythmia induced by psychotropic medications: A proposal for clinical management. European Heart Journal, 35(20), 1306–1315. https://doi.org/10.1093/eurheartj/ehu100
4. Beach, S. R., Celano, C. M., Sugrue, A. M., Adams, C., Ackerman, M. J., Noseworthy, P. A., & Huffman, J. C. (2018). QT Prolongation, Torsades de Pointes, and Psychotropic Medications: A 5-Year Update. Psychosomatics, 59(2), 105–122. https://doi.org/10.1016/j.psym.2017.10.009
5. Drew, B. J., Ackerman, M. J., Funk, M., Gibler, W. B., Kligfield, P., Menon, V., Philippides, G. J., Roden, D. M., & Zareba, W. (2010). Prevention of Torsade de Pointes in Hospital Settings. Circulation, 121(8), 1047–1060. https://doi.org/10.1161/CIRCULATIONAHA.109.192704
6. Food, & Administration, D. (Updated November 3, 2025). Amitriptyline Hydrochloride Tablets. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=33807ee6-8443-4613-8bbe-d66f6010fc60&type=pdf
7. Hicks, J., Sangkuhl, K., Swen, J., Ellingrod, V., Müller, D., Shimoda, K., Bishop, J., Kharasch, E., Skaar, T., Gaedigk, A., Dunnenberger, H., Klein, T., Caudle, K., & Stingl, J. (2017). Clinical pharmacogenetics implementation consortium guideline (CPIC) for CYP2D6 and CYP2C19 genotypes and dosing of tricyclic antidepressants: 2016 update. Clinical Pharmacology & Therapeutics, 102(1), 37–44. https://doi.org/10.1002/cpt.597
8. Hiemke, C., Bergemann, N., Clement, H. W., Conca, A., Deckert, J., Domschke, K., Eckermann, G., Egberts, K., Gerlach, M., Greiner, C., Gründer, G., Haen, E., Havemann-Reinecke, U., Hefner, G., Helmer, R., Janssen, G., Jaquenoud, E., Laux, G., Messer, T., … Baumann, P. (2018). Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology: Update 2017. Pharmacopsychiatry, 51(1–02), 9–62. https://doi.org/10.1055/s-0043-116492
9. By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. (2023). American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. Journal of the American Geriatrics Society, 71(7), 2052–2081. https://doi.org/10.1111/jgs.18372
10. Richelson, E. (1994). Pharmacology of Antidepressants—Characteristics of the Ideal Drug. Mayo Clinic Proceedings, 69(11), 1069–1081. https://doi.org/10.1016/S0025-6196(12)61375-5
11. Roose, S. P., Glassman, A. H., Siris, S. G., Walsh, B. T., Bruno, R. L., & Wright, L. B. (1981). Comparison of imipramine- and nortriptyline-induced orthostatic hypotension: A meaningful difference. Journal of Clinical Psychopharmacology, 1(5), 316–319. https://doi.org/10.1097/00004714-198109000-00008
12. Glassman, A. H., Bigger, J. T., Giardina, E. V., Kantor, S. J., Perel, J. M., & Davies, M. (1979). Clinical characteristics of imipramine-induced orthostatic hypotension. Lancet, 1(8114), 468–472. https://doi.org/10.1016/s0140-6736(79)90824-9
13. Serretti, A., & Mandelli, L. (2010). Antidepressants and body weight: A comprehensive review and meta-analysis. The Journal of Clinical Psychiatry, 71(10), 1259–1272. https://doi.org/10.4088/JCP.09r05346blu
14. Hiemke, C., Bergemann, N., Clement, H., Conca, A., Deckert, J., Domschke, K., Eckermann, G., Egberts, K., Gerlach, M., Greiner, C., Gründer, G., Haen, E., Havemann-Reinecke, U., Hefner, G., Helmer, R., Janssen, G., Jaquenoud, E., Laux, G., Messer, T., … Baumann, P. (2018). Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology: Update 2017. Pharmacopsychiatry, 51(01/02), 9–62. https://doi.org/10.1055/s-0043-116492
15. Ray, W. A., Meredith, S., Thapa, P. B., Hall, K., & Murray, K. T. (2004). Cyclic antidepressants and the risk of sudden cardiac death. Clinical Pharmacology and Therapeutics, 75(3), 234–241. https://doi.org/10.1016/j.clpt.2003.09.019
16. Moraczewski, J., Awosika, A. O., & Aedma, K. K. (2023). Tricyclic Antidepressants. In StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/books/NBK557791/
17. Woolf, A. D., Erdman, A. R., Nelson, L. S., Caravati, E. M., Cobaugh, D. J., Booze, L. L., Wax, P. M., Manoguerra, A. S., Scharman, E. J., Olson, K. R., Chyka, P. A., Christianson, G., & Troutman, W. G. (2007). Tricyclic antidepressant poisoning: An evidence-based consensus guideline for out-of-hospital management. Clinical Toxicology, 45(3), 203–233. https://doi.org/10.1080/15563650701226192
18. Boehnert, M. T., & Lovejoy, F. H. (1985). Value of the QRS Duration versus the Serum Drug Level in Predicting Seizures and Ventricular Arrhythmias after an Acute Overdose of Tricyclic Antidepressants. New England Journal of Medicine, 313(8), 474–479. https://doi.org/10.1056/NEJM198508223130804
19. Food, & Administration, D. (Updated November 24, 2021). Clomipramine Hydrochloride Capsules. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=0bf3c4f3-cf70-490d-89d8-9fb84962489e&type=pdf
20. Food, & Administration, D. (Updated November 1, 2023). Nortriptyline Hydrochloride Capsules. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=b1d9582c-742c-91a8-c2b3-bbdb21220470&type=pdf
21. Gillman, P. K. (2007). Tricyclic antidepressant pharmacology and therapeutic drug interactions updated. British Journal of Pharmacology, 151(6), 737–748. https://doi.org/10.1038/sj.bjp.0707253
22. Glassman, A. H., & Bigger, J. T. (1981). Cardiovascular effects of therapeutic doses of tricyclic antidepressants. A review. Archives of General Psychiatry, 38(7), 815–820. https://doi.org/10.1001/archpsyc.1981.01780320095011
23. Coupland, C. A. C., Hill, T., Dening, T., Morriss, R., Moore, M., & Hippisley-Cox, J. (2019). Anticholinergic Drug Exposure and the Risk of Dementia. JAMA Internal Medicine, 179(8), 1084–1093. https://doi.org/10.1001/jamainternmed.2019.0677
24. Richardson, K., Fox, C., Maidment, I., Steel, N., Loke, Y. K., Arthur, A., Myint, P. K., Grossi, C. M., Mattishent, K., Bennett, K., Campbell, N. L., Boustani, M., Robinson, L., Brayne, C., Matthews, F. E., & Savva, G. M. (2018). Anticholinergic drugs and risk of dementia: Case-control study. BMJ, 361, k1315. https://doi.org/10.1136/bmj.k1315
25. Pacher, P., & Kecskemeti, V. (2004). Cardiovascular Side Effects of New Antidepressants and Antipsychotics: New Drugs, old Concerns? Current Pharmaceutical Design, 10(20), 2463–2475. https://doi.org/10.2174/1381612043383872
26. Higgins, A., Nash, M., & Lynch, A. M. (2010). Antidepressant-associated sexual dysfunction: Impact, effects, and treatment. Drug, Healthcare and Patient Safety, 2, 141–150. https://doi.org/10.2147/DHPS.S7634
27. Barbuti, M., Menculini, G., Verdolini, N., Pacchiarotti, I., Kotzalidis, G. D., Tortorella, A., Vieta, E., & Perugi, G. (2023). A systematic review of manic/hypomanic and depressive switches in patients with bipolar disorder in naturalistic settings: The role of antidepressant and antipsychotic drugs. European Neuropsychopharmacology, 73, 1–15. https://doi.org/10.1016/j.euroneuro.2023.04.013
28. Koszewska, I., & Rybakowski, J. K. (2009). Antidepressant-induced mood conversions in bipolar disorder: A retrospective study of tricyclic versus non-tricyclic antidepressant drugs. Neuropsychobiology, 59(1), 12–16. https://doi.org/10.1159/000202824
29. Heinonen, O. P., Slone, D., Monson, R. R., Hook, E. B., & Shapiro, S. (1977). Cardiovascular birth defects and antenatal exposure to female sex hormones. The New England Journal of Medicine, 296(2), 67–70. https://doi.org/10.1056/NEJM197701132960202
30. Altshuler, L. L., & Hendrick, V. C. (1996). Pregnancy and psychotropic medication: Changes in blood levels. Journal of Clinical Psychopharmacology, 16(1), 78–80. https://doi.org/10.1097/00004714-199602000-00015
31. McElhatton, P. R., Garbis, H. M., Eléfant, E., Vial, T., Bellemin, B., Mastroiacovo, P., Arnon, J., Rodríguez-Pinilla, E., Schaefer, C., Pexieder, T., Merlob, P., & Dal Verme, S. (1996). The outcome of pregnancy in 689 women exposed to therapeutic doses of antidepressants. A collaborative study of the European Network of Teratology Information Services (ENTIS). Reproductive Toxicology, 10(4), 285–294. https://doi.org/10.1016/0890-6238(96)00057-3
32. Altshuler, L. L., Cohen, L., Szuba, M. P., Burt, V. K., Gitlin, M., & Mintz, J. (1996). Pharmacologic management of psychiatric illness during pregnancy: Dilemmas and guidelines. The American Journal of Psychiatry, 153(5), 592–606. https://doi.org/10.1176/ajp.153.5.592
33. Brunel, P., Vial, T., Roche, I., Bertolotti, E., & Evreux, J. C. (1994). [Follow-up of 151 pregnant women exposed to antidepressant treatment (MAOI excluded) during organogenesis]. Therapie, 49(2), 117–122. https://www.ncbi.nlm.nih.gov/pubmed/7817334
34. Huybrechts, K. F., Palmsten, K., Avorn, J., Cohen, L. S., Holmes, L. B., Franklin, J. M., Mogun, H., Levin, R., Kowal, M., Setoguchi, S., & Hernandez-Diaz, S. (2014). Antidepressant Use in Pregnancy and the Risk of Cardiac Defects. The New England Journal of Medicine, 370(25), 2397–2407. https://doi.org/10.1056/NEJMoa1312828
35. Nulman, I., Rovet, J., Stewart, D. E., Wolpin, J., Gardner, H. A., Theis, J. G., Kulin, N., & Koren, G. (1997). Neurodevelopment of children exposed in utero to antidepressant drugs. The New England Journal of Medicine, 336(4), 258–262. https://doi.org/10.1056/NEJM199701233360404
36. Nulman, I., Rovet, J., Stewart, D. E., Wolpin, J., Pace-Asciak, P., Shuhaiber, S., & Koren, G. (2002). Child development following exposure to tricyclic antidepressants or fluoxetine throughout fetal life: A prospective, controlled study. The American Journal of Psychiatry, 159(11), 1889–1895. https://doi.org/10.1176/appi.ajp.159.11.1889
37. Food, & Administration, D. (2010). Silenor™ (doxepin) tablets for oral administration. https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/022036lbl.pdf
38. Vieweg, W. V. R., Wood, M. A., Fernandez, A., Beatty-Brooks, M., Hasnain, M., & Pandurangi, A. K. (2009). Proarrhythmic risk with antipsychotic and antidepressant drugs: Implications in the elderly. Drugs & Aging, 26(12), 997–1012. https://doi.org/10.2165/11318880-000000000-00000
39. Liebelt, E. L., Francis, P. D., & Woolf, A. D. (1995). ECG lead aVR versus QRS interval in predicting seizures and arrhythmias in acute tricyclic antidepressant toxicity. Annals of Emergency Medicine, 26(2), 195–201. https://doi.org/10.1016/s0196-0644(95)70151-6
40. Monteban-Kooistra, W. E., van den Berg, M. P., Tulleken, J. E., Ligtenberg, J. J. M., Meertens, J. H. J. M., & Zijlstra, J. G. (2006). Brugada electrocardiographic pattern elicited by cyclic antidepressants overdose. Intensive Care Medicine, 32(2), 281–285. https://doi.org/10.1007/s00134-005-0007-3
41. Ramasubbu, B., James, D., Scurr, A., & Sandilands, E. A. (2016). Serum alkalinisation is the cornerstone of treatment for amitriptyline poisoning. BMJ Case Reports, 2016, 10.1136/bcr-2016-214685. https://doi.org/10.1136/bcr-2016-214685
42. Hultén, B. A., Adams, R., Askenasi, R., Dallos, V., Dawling, S., Heath, A., & Volans, G. (1988). Activated charcoal in tricyclic antidepressant poisoning. Human Toxicology, 7(4), 307–310. https://doi.org/10.1177/096032718800700402
43. Pentel, P. R., & Benowitz, N. L. (1986). Tricyclic antidepressant poisoning. Management of arrhythmias. Medical Toxicology, 1(2), 101–121. https://doi.org/10.1007/BF03259831
44. Pentel, P., & Peterson, C. D. (1980). Asystole complicating physostigmine treatment of tricyclic antidepressant overdose. Annals of Emergency Medicine, 9(11), 588–590. https://doi.org/10.1016/s0196-0644(80)80232-0



