In a nutshell
Tricyclic antidepressants (TCAs) are second- or third-line agents for depression, typically reserved for SSRI/SNRI failure or when comorbid conditions favor their use. Secondary amines (nortriptyline, desipramine, protriptyline) are better tolerated than tertiary amines (amitriptyline, clomipramine, doxepin, imipramine, trimipramine). Amitriptyline has the highest efficacy signal among all antidepressants, but its tolerability is limited. This guide covers indications, agent selection, dosing, and titration; for safety, monitoring, drug interactions, and overdose management, see Prescribing Tricyclic Antidepressants Safely.

- Consider TCAs when:
- Depression has not responded to SSRIs/SNRIs (second- or third-line option)
- Melancholic depression (some evidence favoring TCAs over SSRIs)
- Comorbid chronic pain syndromes: neuropathic pain, migraine, fibromyalgia, or chronic headache (amitriptyline first-line for neuropathic pain; NNT 3.6)
- OCD refractory to SSRIs: Clomipramine is FDA-approved for OCD; efficacy comparable to SSRIs but used second-line due to side-effect burden
- Comorbid insomnia: Low-dose doxepin 3–6 mg is FDA-approved; low-dose amitriptyline is used off-label
- Prefer alternatives when:
- High suicide risk without adequate safety monitoring (overdose lethality)
- Older adults
- Beers Criteria: TCAs listed as potentially inappropriate due to anticholinergic burden and fall risk
- If required, use nortriptyline or desipramine
- Cardiac disease, conduction abnormalities (QRS >100 ms, bundle branch block, contraindicated during acute MI recovery)
- Patients taking strong CYP2D6 inhibitors (fluoxetine, paroxetine, bupropion)
- Can increase TCA levels 200–800%
- Bipolar disorder (TCAs carry the highest mania switch rate among antidepressant classes)
- Borderline personality disorder: Risk of impulsive overdose, aggression, disinhibition

- Key clinical recommendations:
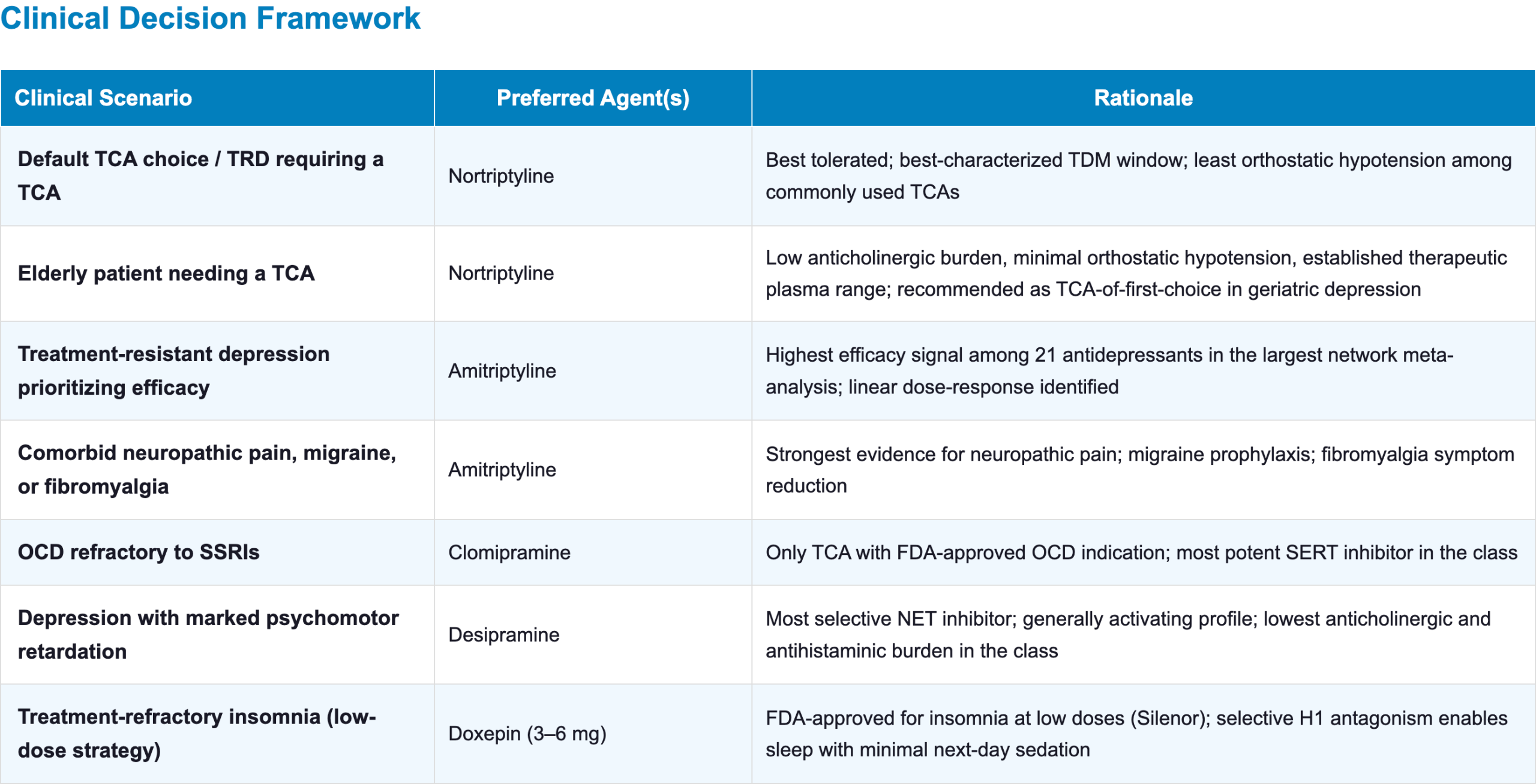
- Default TCA choice: Nortriptyline
- Best tolerated, best-characterized therapeutic window (70–170 ng/mL)
- The least orthostatic hypotension among commonly used TCAs
- Prefer secondary amines (particularly nortriptyline, desipramine) over tertiary amines when possible.
- Fewer anticholinergic, sedative, and cardiovascular effects
- Therapeutic drug monitoring (TDM):
- Strongly recommended for all TCAs; draw trough levels at steady state
- Baseline ECG:
- Obtain for all patients ≥40 years
- Consider for younger patients with cardiac history or concomitant QT-prolonging drugs
- Default TCA choice: Nortriptyline
Pharmacodynamics and mechanism of action
- TCAs exert their antidepressant effect primarily by inhibiting serotonin (SERT) and norepinephrine (NET) reuptake [1,2]
- Additional blockade of H1, M1, and α1 receptors accounts for their characteristic side-effect profile and some therapeutic properties
- The relative potency of serotonin vs norepinephrine reuptake inhibition varies by agent and defines the tertiary/secondary amine distinction
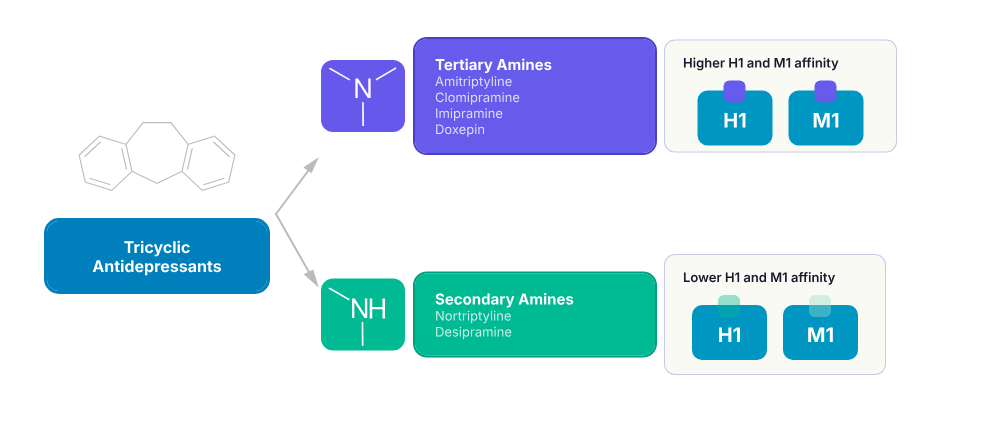

- Tertiary amines (amitriptyline, clomipramine, doxepin, imipramine, trimipramine)
- Balanced or serotonin-dominant SERT/NET inhibition [1,3]
- Higher affinity for histamine H1 receptors (sedation, weight gain) and muscarinic M1 receptors (anticholinergic effects)
- Two methyl groups on the nitrogen atom of the side chain
- Clomipramine is the most serotonergic TCA; its capacity to inhibit serotonin reuptake is thought to explain its efficacy in OCD [4]
- Its active metabolite, desmethylclomipramine (DMI), preferentially inhibits NE reuptake, creating a dual serotonergic/noradrenergic profile

- Secondary amines (nortriptyline, desipramine, protriptyline)
- Preferential NET inhibition [1]
- Lower H1 and M1 affinity (less sedation, fewer anticholinergic effects, less weight gain)
- Loss of one methyl group shifts affinity toward NET and reduces ancillary receptor binding
- Desipramine: the least antihistaminic of all TCAs and among the least anticholinergic (along with nortriptyline and protriptyline)
- Nortriptyline and desipramine are active (demethylated) metabolites of amitriptyline and imipramine, respectively [5]
- Additional off-target receptor blockade
- H1 (histamine) antagonism
- Sedation, increased appetite, weight gain [1]
- Strongest: doxepin (highest H1 affinity of any TCA) > amitriptyline
- Weakest: desipramine, nortriptyline
- Clinical pearl: Doxepin’s extreme H1 affinity is exploited therapeutically at ultra-low doses (3–6 mg) for sleep maintenance insomnia (FDA-approved)
- M1 (muscarinic) antagonism
- Dry mouth, blurred vision, constipation, urinary retention, cognitive impairment, tachycardia [1,6]
- More pronounced with tertiary amines
- α1 (adrenergic) antagonism
- Orthostatic hypotension, reflex tachycardia, dizziness [1]
- More pronounced with tertiary amines
- Major risk factor for falls and syncope in older adults
- Sodium channel blockade[6,7]
- TCAs block voltage-gated sodium channels in myocardial cells, primarily slowing the rapid influx of sodium ions during phase 0 of the cardiac action potential
- Also contributes to the analgesic effect in neuropathic pain by stabilizing neuronal membranes and reducing ectopic firing
- Clinical implications:
- QRS widening thresholds
- Cardiac toxicity
- Overdose management
- hERG potassium channel blockade[8]
- QTc prolongation risk via delayed repolarization
- H1 (histamine) antagonism
Indications
Depression
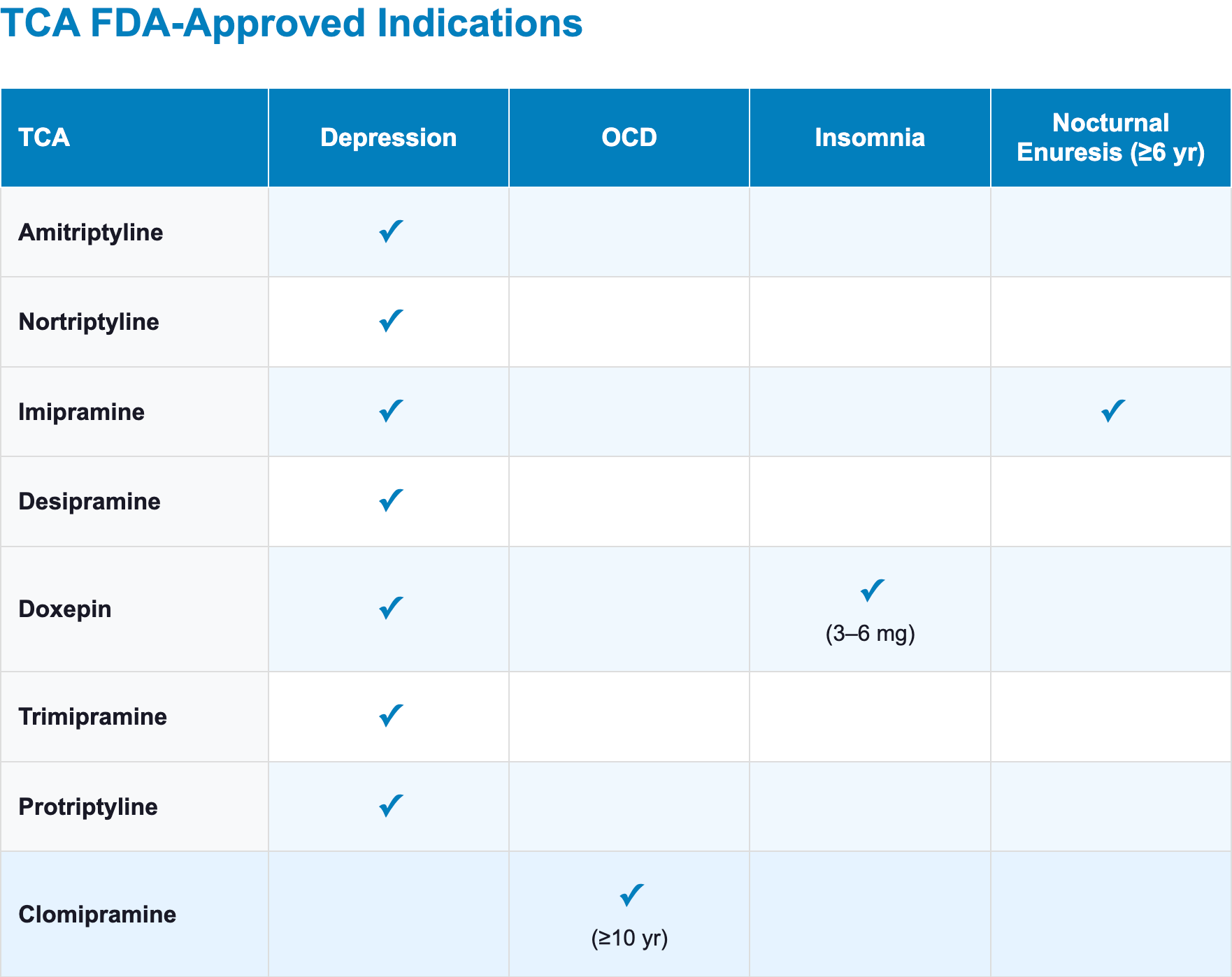
- FDA-approved for depression: amitriptyline, nortriptyline, imipramine, desipramine, doxepin, trimipramine, protriptyline
- Clomipramine is widely used off-label for depression, particularly in Europe, though its only FDA approval is for OCD [4]
- FDA labeling language for amitriptyline and nortriptyline notes that “endogenous depression” (severe, melancholic-type) “is more likely to be alleviated” [6,7]
- Efficacy evidence:
- Amitriptyline shows the highest efficacy point estimate (OR 2.13, 95% CrI 1.89–2.41) among 21 antidepressants studied in the largest network meta-analysis [9]
- Head-to-head trials show no overall efficacy difference between TCAs and SSRIs [10]
- Clinical positioning:
- Second- or third-line antidepressant after SSRI/SNRI failure or intolerance; may be of choice when comorbid conditions favor TCA use (pain, insomnia, OCD)
- Melancholic depression
- In a meta-analysis comparing drug classes, all patients (melancholic and non-melancholic) responded better to TCAs than SRIs, but this difference did not preferentially favor melancholic depression [11]
- A TCA trial may be considered after SSRI/SNRI failure in patients with melancholic features
Obsessive-compulsive disorder
- Clomipramine is the only TCA with an FDA-approved indication for OCD (ages ≥10 years) [4,12]
- SSRIs are preferred as first-line pharmacotherapy for OCD due to their similar efficacy and superior tolerability
- Clomipramine is recommended as second-line after SSRI failure or when the patient has a prior good response to it [13–15]
- Dosing for OCD [4]
- Starting dose: 25 mg/day;
- Target dose: 100–200 mg/day
- Maximum 250 mg/day (adults); 3 mg/kg/day or 200 mg/day (children/adolescents, whichever is less)
- As adjunct to SSRI in treatment-resistant OCD: 25–75 mg added to ongoing SSRI; monitor for serotonergic side effects [13,16]
Comorbid pain conditions
- Neuropathic pain
- TCAs are recommended as first-line agents for neuropathic pain (alongside gabapentinoids, duloxetine, and venlafaxine) in international guidelines [17]
- Amitriptyline and nortriptyline are the most widely studied agents; the NNT for TCAs to achieve at least moderate pain relief vs. placebo is 3.6 [17–19]
- Doses used for neuropathic pain are lower than antidepressant doses [17]
- Amitriptyline 25–75 mg/day
- Nortriptyline 25–100 mg/day
- Migraine prophylaxis
- Amitriptyline is the most widely studied TCA for migraine prevention and is recommended by multiple headache guidelines [20,21]
- Effective doses are typically 10–75 mg at bedtime; efficacy emerges over 4–8 weeks [20,21]
- Chronic tension-type headache:
- Amitriptyline has shown efficacy in chronic tension-type headache prevention [20,22]
- Stress management combined with amitriptyline was more effective than either treatment alone
- Fibromyalgia
- Amitriptyline and nortriptyline are among the TCAs with the strongest evidence for fibromyalgia symptom improvement [23]
- Low-dose amitriptyline (10–50 mg QHS) has been shown to reduce pain intensity and improve sleep quality in fibromyalgia [23,24]
- IBS-D (diarrhea-predominant IBS)
- Low-dose TCAs are effective for IBS-D, reducing abdominal pain and stool frequency [25–27]
- Amitriptyline 10–30 mg at bedtime
- Anticholinergic properties of tertiary amines slow colonic transit, providing a therapeutic “side effect” in IBS-D
- For IBS-C, secondary amines (nortriptyline, desipramine) are preferred to avoid exacerbating constipation [25]
- Low-dose TCAs are effective for IBS-D, reducing abdominal pain and stool frequency [25–27]
Other indications
- Insomnia:
- Doxepin 3–6 mg (Silenor) is FDA-approved for the treatment of insomnia characterized by difficulty with sleep maintenance [28]
- At these doses, it selectively targets H1 receptors with minimal anticholinergic or adrenergic effects [29]
- Not recommended in patients with severe sleep apnea [28]
- Low-dose amitriptyline (10–25 mg QHS) is used off-label for insomnia, though RCT evidence for this practice is limited [30,31]
- Doxepin 3–6 mg (Silenor) is FDA-approved for the treatment of insomnia characterized by difficulty with sleep maintenance [28]
- Panic disorder:
- Imipramine has the strongest historical evidence base for panic disorder among TCAs; clomipramine is also effective [32]
- TCAs are typically reserved for second-line use after SSRI/SNRI failure due to their tolerability profile [14]
- PTSD:
- Imipramine and amitriptyline have shown some efficacy in PTSD, but evidence is limited and of low quality; they are not considered first-line [12,33]
- Nocturnal enuresis in children ≥6 years:
- Imipramine is the only TCA that has been FDA-approved for this indication [34]
- TCAs reduce the number of wet nights during treatment, but the effect is not sustained after discontinuation, and most children relapse [35]
- Smoking cessation:
- Nortriptyline is a second-line agent for smoking cessation; not FDA-approved for this indication [36,37]
- Cataplexy in narcolepsy:
- Clomipramine is used off-label for cataplexy due to its potent serotonergic and noradrenergic reuptake inhibition [31]
When to prescribe each TCA
- Secondary amines (nortriptyline, desipramine) generally have a more favorable side effect profile compared with tertiary amines (amitriptyline, clomipramine, doxepin, imipramine, trimipramine) [1,38]
- Nortriptyline and desipramine have the most pharmacologically desirable characteristics, with fewer off-target interactions and safer profiles in combination therapy [38]
Agent profiles
Nortriptyline
- Best-tolerated, most versatile TCA.
- Consider for:
- Most patients requiring a TCA (default choice)
- Older adults, if a TCA is needed [39]
- Patients requiring precise dose titration with therapeutic drug monitoring (TDM)
- Breastfeeding mothers requiring a TCA — low breast milk excretion and minimal infant effects [40]
- Key advantages:
- Among the least anticholinergic TCA [1,41–43]
- Less sedation and significantly less orthostatic hypotension than imipramine or amitriptyline [43]
- Best-characterized therapeutic window, which enables reliable TDM-guided dosing [44]
- Nortriptyline is the only antidepressant with a well-defined inverted-U shaped concentration–response curve [44]
- Patients are less likely to respond if levels are too low or too high [45]
- As a secondary amine, dependent on only CYP2D6 for inactivation (not CYP2C19), drug interactions are more predictable [38,46]
- No active metabolites of major concern, and no significant CYP450 enzyme inhibition in the therapeutic dose range [38]
- Key limitation:
- Lower serotonergic activity than tertiary amines [38,41]
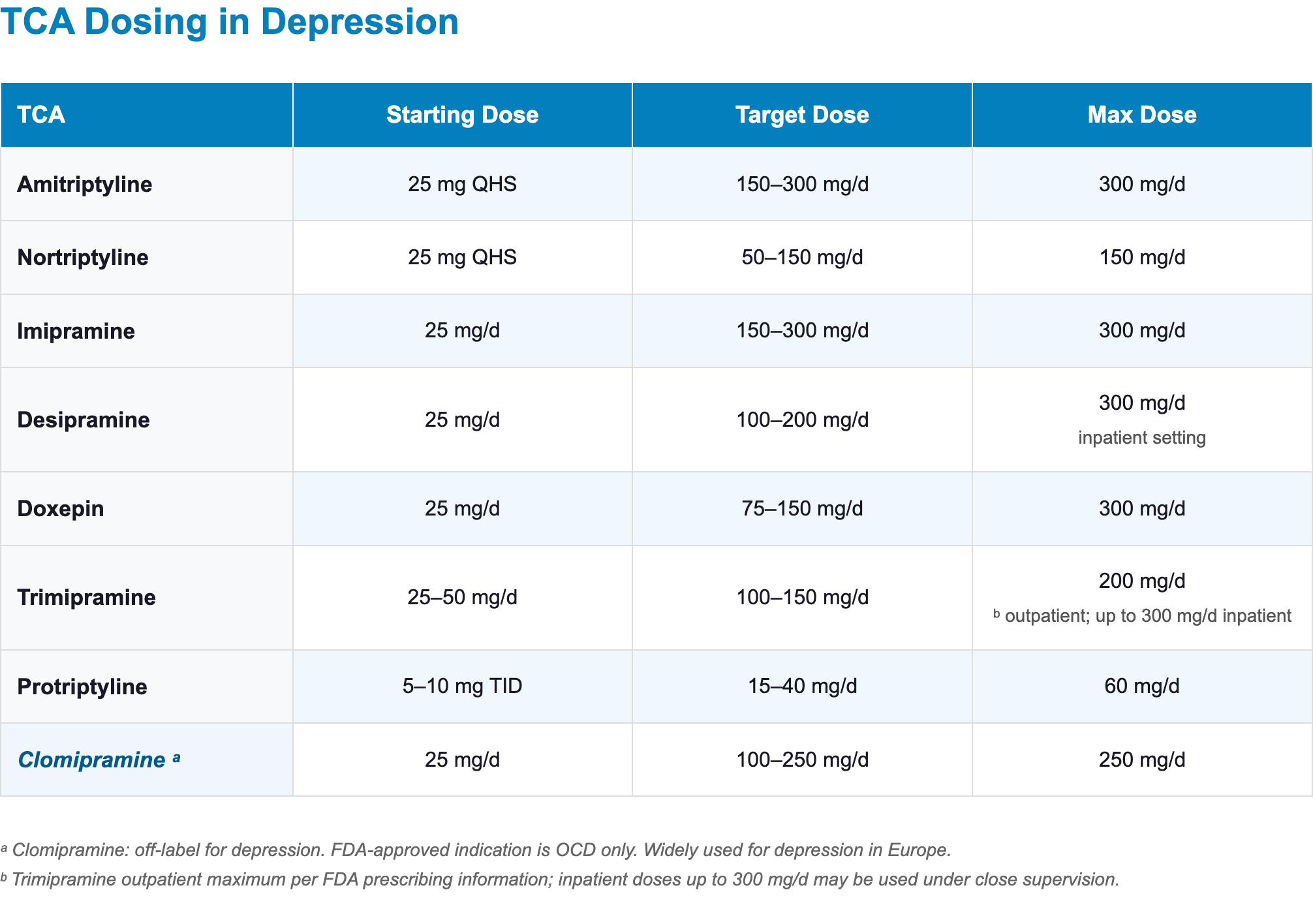
- Dosing:
- Starting dose: 25 mg at bedtime
- Target dose: 50–150 mg/day
- Maximum dose: 150 mg/day (per FDA label; doses above 150 mg/day are not recommended) [7]
- Check serum levels to guide dose optimization, especially above 100 mg/day [7,44]
Amitriptyline
- Highest efficacy, most side effects [1,9]
- Consider for:
- Treatment-resistant depression
- Comorbid neuropathic pain
- Migraine prophylaxis
- Fibromyalgia
- IBS-D
- Comorbid insomnia (when sedation is desired)
- Key advantages:
- Ranked highest for efficacy among 21 antidepressants in a landmark network meta-analysis [9]
- A dose-response meta-analysis of 602 RCTs reported a linear dose-response relationship for amitriptyline and clomipramine (unlike most antidepressants, which reach maximum efficacy at lower or middle doses) [47]
- Key limitations:
- Tolerability is limited by its side-effect burden [9]
- Most anticholinergic and among the most sedating TCAs [1,41]
- Highest orthostatic hypotension risk together with imipramine, correlated with potent alpha-1 adrenergic blockade [42]
- In 89 head-to-head trials, no overall difference in response rates was detected between TCAs and SSRIs; the absolute efficacy advantage does not consistently translate to clinical practice [10]
- TDM target: amitriptyline + nortriptyline (combined) = 80–200 ng/mL; alert level: 300 ng/mL [44]
- Dosing:
- Starting dose: 25 mg at bedtime, increase by 25–50 mg every 5–7 days
- Target dose: 150–300 mg/day
- Maximum dose: 300 mg/day
- Elderly: start lower (e.g., 10–25 mg at bedtime) and titrate slowly [6]
- Migraine prophylaxis: 10–75 mg
- Fibromyalgia: 25–50 mg
Clomipramine
- OCD and serotonergic-dominant profile
- Consider for
- OCD refractory to SSRIs (only TCA with FDA-approved OCD indication) [4]
- Depression requiring strong serotonergic activity; cataplexy (off-label) [4]
- Key advantages:
- Most potent serotonin reuptake inhibitor in the TCA class [4,38]
- Active metabolite desmethylclomipramine adds noradrenergic coverage, creating an SNRI-like dual profile
- Efficacy is comparable to SSRIs in network meta-analysis [9]
- Key limitations:
- Tolerability, not efficacy, is its limiting factor; highest all-cause dropout rate among antidepressants in network meta-analysis [9]
- Seizure risk higher at doses >250 mg/day [4]
- Careful dose titration required due to nonlinear pharmacokinetics above 150 mg/day [4]
- Drug-induced liver injury highest in the TCA class; consider periodic liver function monitoring [4]
- TDM target: clomipramine + desmethylclomipramine (combined) = 230–450 ng/mL [44]
- Dosing:
- Starting dose: 25 mg/day; divide doses with meals during initial titration to reduce GI side effects
- Target dose: increase to ~100 mg/day over 2 weeks, then gradually to 200–250 mg/day over several additional weeks
- Maximum dose: 250 mg/day (per FDA label; linked to seizure risk above this threshold) [4]
Imipramine
- FDA-approved for nocturnal enuresis in children ≥6 years [34]
- Dose-response relationship: 300 mg of imipramine/desipramine was significantly more effective than 150 mg in a meta analysis [48]
- The active metabolite desipramine contributes a favorable noradrenergic profile
- Among the highest orthostatic hypotension risk in the TCA class [8,43]
- TDM target: imipramine + desipramine (combined) 175–300 ng/mL [44]
- Dosing:
- Starting dose: 25 mg/day
- Target dose: 150–300 mg/day
- Maximum dose: 300 mg/day
Desipramine
- Most noradrenergic and least anticholinergic TCA [1,38,41]
- Generally activating profile, though some patients find it sedating
- Less anticholinergic burden and weight gain potential
- Off-label uses include ADHD and functional dyspepsia
- As a secondary amine, dependent on only CYP2D6 for inactivation (not CYP2C19), drug interactions are more predictable [38,46]
- Key safety concerns:
- Appears more toxic in overdose than other TCAs [8]
- Reports of sudden death in children treated with desipramine have prompted particular caution in pediatric use [49,50]
- Intense noradrenergic activity may cause sinus tachycardia [38]
- TDM target: 100–300 ng/mL [44]
- Dosing:
- Starting dose: 25 mg/day
- Target dose: 100–200 mg/day
- Maximum dose: 300 mg/day (inpatient setting)
Doxepin
- Highest H1 receptor affinity: most potent antihistamine in the TCA class [1,38]
- At low dose (3–6 mg as Silenor):
- FDA-approved for treatment of insomnia characterized by difficulty with sleep maintenance [28]
- At this dose it functions as a selective H1 antagonist without engaging muscarinic, alpha-adrenergic, or monoamine reuptake mechanisms at clinically meaningful levels [28]
- At antidepressant doses (75–150 mg):
- Very sedating, significant weight gain
- TDM target: doxepin + nordoxepin (combined) = 50–150 ng/mL [44]
- Dosing:
- Starting dose: 25 mg/day
- Target dose: 75–150 mg/day
- Maximum dose: 300 mg/day (hospitalized patients under close supervision)
Trimipramine
- Rarely prescribed in contemporary practice
- Weakest norepinephrine reuptake inhibition of all TCAs; mechanistically closer to a sedating antihistamine than a classical reuptake inhibitor [38,41]
- Unlike other TCAs, trimipramine was shown to improve sleep efficiency and continuity vs. placebo while preserving REM architecture [51]
- Potent H1, M1, and alpha-1 blockade may produce significant sedation, anticholinergic burden, orthostatic hypotension [1]
- Dosing:[52]
- Starting dose: 25–50 mg/day (or 75 mg/day in divided doses; lower starting doses used in elderly or sensitive patients)
- Target dose: 100–200 mg/day
- Maximum dose: 200 mg/day (outpatient); 300 mg/day (inpatient, under supervision)
Protriptyline
- Rarely prescribed; available only in the United States [53]
- Protriptyline has the highest NET/SERT selectivity ratio, producing a markedly stimulating noradrenergic profile [41]
- Most consistently activating TCA
- Should not be dosed at bedtime
- Usually given in divided doses TID–QID
- Lowest H1 receptor affinity among TCAs: minimal sedation and weight gain potential [1]
- FDA-approved for depression only; also used off-label for narcolepsy, ADHD, and headache [53]
- Pharmacokinetics: Exceptionally long half-life (~74 h; range 54–92 h). Up to 4 weeks to reach steady state; slower titration required [54]
- Cardiovascular profile: Most likely among TCAs to cause tachycardia and agitation in clinical use [53]
- Dosing:[53]
- Starting dose: 5–10 mg TID (15–30 mg/day total)
- Target dose: 15–40 mg/day
- Maximum dose: 60 mg/day
- ECG monitoring recommended in patients >60 years or at doses >20 mg/day
Therapeutic Drug Monitoring, Dosing, and Titration
Therapeutic drug monitoring
- Therapeutic drug monitoring (TDM) is strongly recommended for all TCAs [44]
- Level 1 (strongly recommended): amitriptyline, clomipramine, nortriptyline, imipramine
- Level 2 (recommended): doxepin, desipramine
- Draw trough levels 12 hours (±2 h) after the last dose, at steady state (after 1–2 weeks at a stable dose) [44]
- For tertiary amines, always measure parent compound + active metabolite together (e.g., amitriptyline + nortriptyline) [44]
- Indications for TDM beyond routine baseline:
- After reaching steady state (≥5 days after dose change; longer in elderly)
- After dose changes or changes in CYP2D6/CYP2C19 inhibitors/inducers
- Suspected non-adherence or non-response at adequate doses
- Signs of toxicity or poor tolerability
- CYP2D6/CYP2C19 poor or ultrarapid metabolizers [44,46]
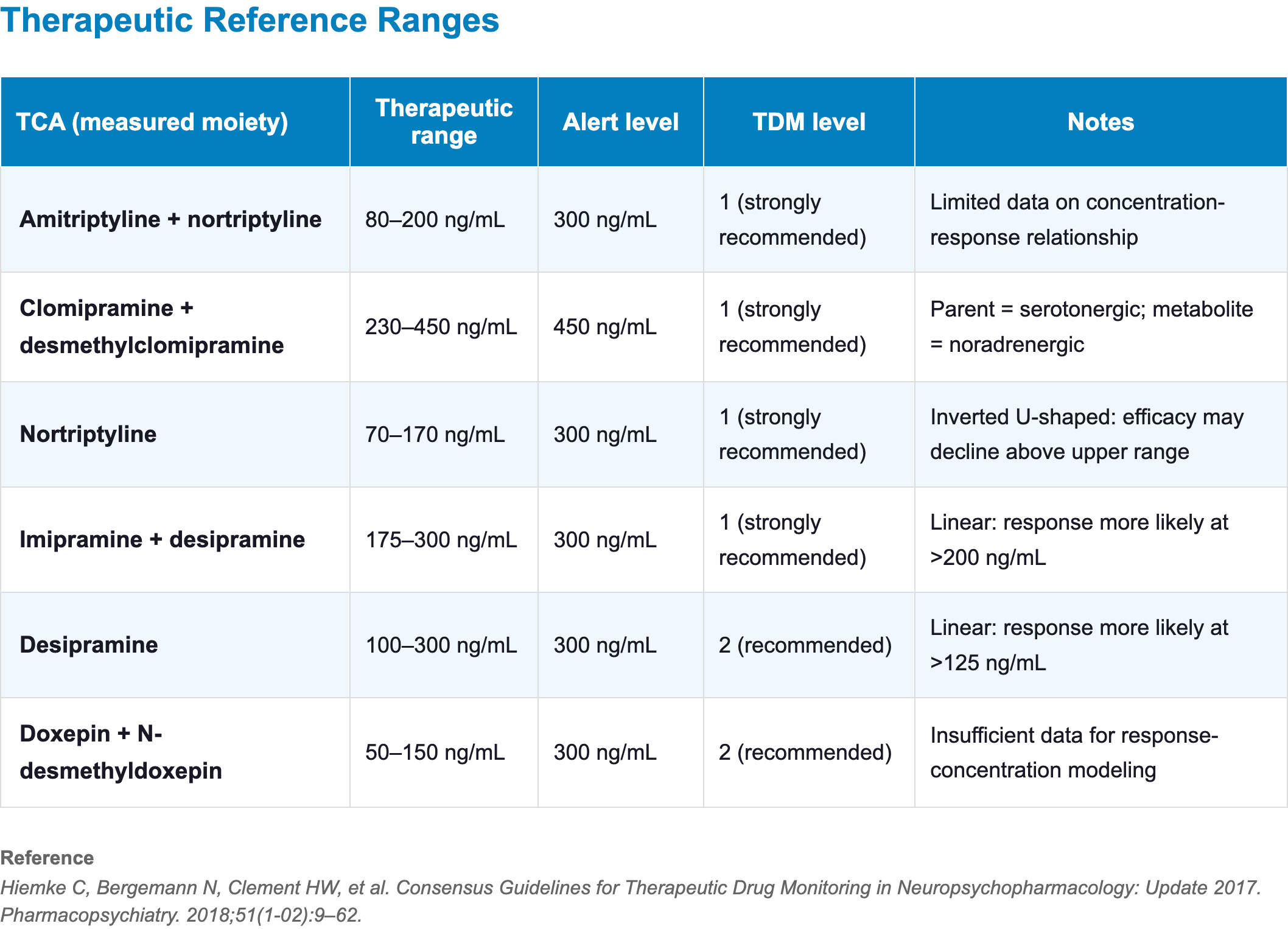
- Nortriptyline has the best-established concentration–response relationship among all TCAs
- It exhibits a “therapeutic window” where levels above the upper range may show decreased efficacy (not just increased toxicity), making TDM particularly valuable for this agent [44]
- The FDA label cites a therapeutic range of 50–150 ng/mL for nortriptyline [7], while the AGNP guideline (2017) recommends 70–170 ng/mL based on broader meta-analytic evidence [44]
Half-life
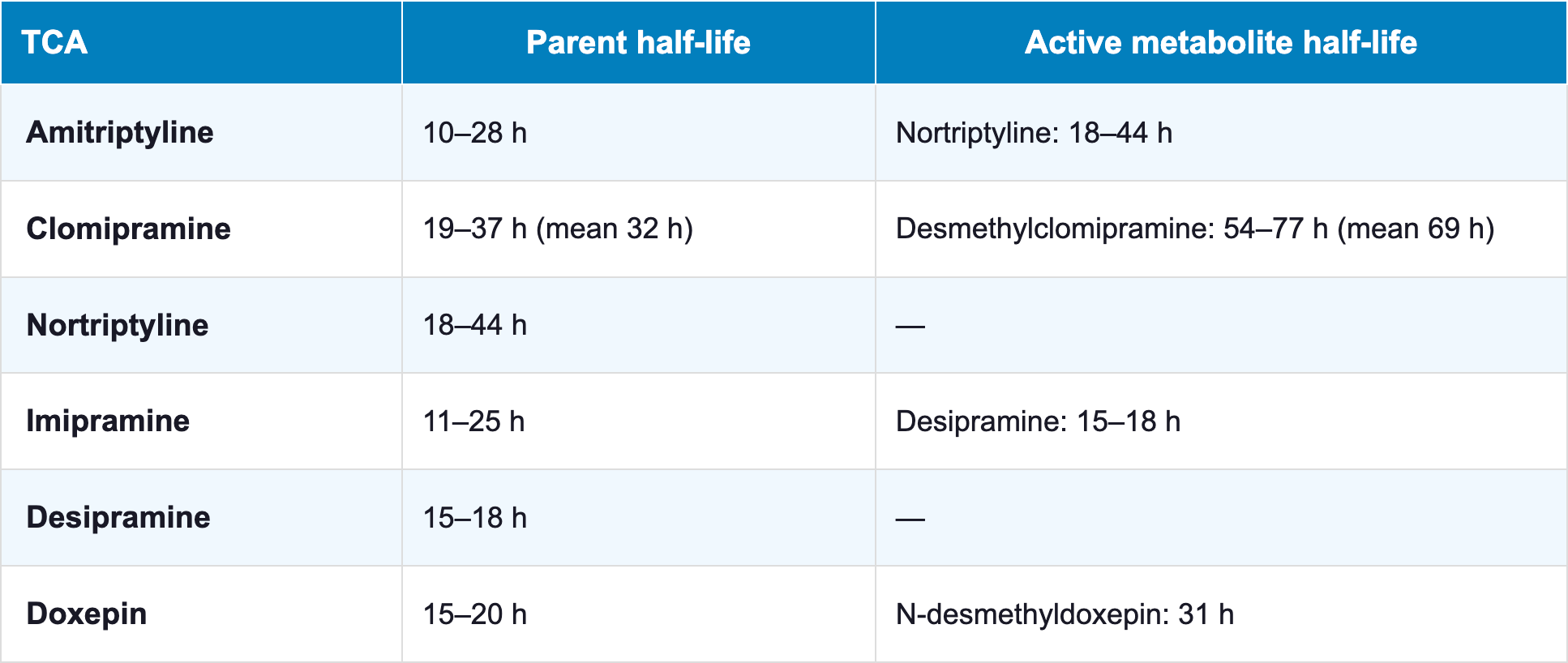
- All above TCAs can be given once daily at bedtime after initial titration
- Long half-lives support once-daily dosing [4,6,7]
- Steady state: 7–14 days for most TCAs; 2–3 weeks for clomipramine given the long desmethylclomipramine half-life [4]
Dosage forms
- Amitriptyline
- Tablets
- 10 mg, 25 mg, 50 mg, 75 mg, 100 mg, 150 mg
- Generic (Elavil discontinued)
- Tablets
- Nortriptyline
- Capsules
- 10 mg, 25 mg, 50 mg, 75 mg
- Generic (Pamelor discontinued)
- Oral solution
- 10 mg/5 mL
- Generic
- Capsules
- Clomipramine
- Capsules
- 25 mg, 50 mg, 75 mg
- Generic, Anafranil
- Capsules
- Imipramine
- Tablets (hydrochloride salt)
- 10 mg, 25 mg, 50 mg
- Generic (Tofranil)
- Capsules (pamoate salt)
- 75 mg, 100 mg, 125 mg, 150 mg
- Generic (Tofranil-PM)
- Tablets (hydrochloride salt)
- Desipramine
- Tablets
- 10 mg, 25 mg, 50 mg, 75 mg, 100 mg, 150 mg
- Generic (Norpramin discontinued)
- Tablets
- Doxepin
- Capsules (antidepressant doses)
- 10 mg, 25 mg, 50 mg, 75 mg, 100 mg, 150 mg
- Generic, Sinequan (discontinued)
- Tablets (low-dose, insomnia indication)
- 3 mg, 6 mg
- Silenor
- Not interchangeable with antidepressant-dose formulations
- Capsules (antidepressant doses)
Formulation tips
- Generics
- All TCAs are available as generics; original brand names have largely been discontinued in the US market
- Bedtime dosing
- All TCAs can be administered once daily at bedtime after initial titration
- Exception: Protriptyline, which tends to be activating
- Bedtime dosing minimizes daytime sedation and leverages the sedative properties of many agents
- Patients who cannot tolerate a single bedtime dose may divide into 2–3 doses with the larger portion at bedtime
- All TCAs can be administered once daily at bedtime after initial titration
- Clomipramine titration
- During initial titration, divided doses with meals are recommended to reduce gastrointestinal side effects; once-daily bedtime dosing can be adopted after titration
- Oral solution
- Nortriptyline oral solution (10 mg/5 mL) is useful for fine dose titration in older adults, those with swallowing difficulties, or when small dose adjustments are needed for TDM-guided optimization
- Capsules:
- Imipramine pamoate (75–150 mg) allows higher single-dose administration, which may simplify adherence for patients requiring doses above 50 mg
- Non-oral formulations
- Parenteral formulations (intramuscular amitriptyline, imipramine, and clomipramine; intravenous clomipramine) are available in some countries outside the United States [55]
Switching To and From TCAs
Switching from SSRI/SNRI to TCA
- Most SSRIs/SNRIs (non-fluoxetine, non-paroxetine):
- Cross-taper over 2–4 weeks; gradually reduce SSRI/SNRI while introducing TCA at a low starting dose (typically 25 mg) and titrating slowly [56,57]
- From fluoxetine:
- Taper and stop fluoxetine (or stop directly if dose ≤40 mg/day), wait at least 7–14 days, then start TCA at 25 mg and maintain this low dose for a further 3 weeks before titrating [56]
- Fluoxetine inhibits CYP2D6 and its active metabolite norfluoxetine (t½ ~4–16 days) sustains this inhibition for weeks after stopping;
- TCA plasma levels can increase 2- to 10-fold, and this effect persists 3 weeks or longer after fluoxetine discontinuation [58]
- From paroxetine:
- Taper and stop; CYP2D6 inhibition can persist 4–6 weeks after long-term use, elevating TCA levels well beyond the discontinuation date [59]
- Start TCA at reduced dose (25 mg) and use TDM guidance during titration
- Counsel patients
- During the transition period they may experience temporary symptoms from discontinuation of the first drug (restlessness, nausea, sleep disturbance) before the TCA reaches therapeutic levels
Switching from TCA to SSRI/SNRI:
- Most TCAs (non-clomipramine):
- Gradually reduce TCA dose by 50%, start SSRI at standard starting dose, then taper TCA over 2–4 further weeks while the SSRI is titrated upward [56,57]
- Paroxetine and fluoxetine (CYP2D6 inhibitors) will elevate remaining TCA levels during the overlap, even as the TCA is being tapered; account for this in timing and starting dose [56]
- From clomipramine:
- Taper and stop clomipramine completely before starting an SSRI or SNRI [56]
- Cross-taper is not recommended due to clomipramine’s potent SERT inhibition (combining with an SSRI may increase serotonin syndrome risk) [38,60]
- Discontinuation:
- Taper all TCAs by 25–50% every 1–2 weeks to minimize cholinergic rebound and withdrawal symptoms (nausea, headache, irritability, sleep disturbance) [4,6,7]
MAOI washout protocols
- Any TCA to MAOI:
- A minimum 14-day washout is required after stopping the TCA before starting an MAOI
- 21 days recommended for clomipramine given the long desmethylclomipramine half-life (54–77 h)
- MAOI to any TCA:
- A 14-day washout is required after stopping the MAOI before starting a TCA [4,6,7]
- Cross-taper between TCAs and MAOIs is contraindicated (risk of serotonin syndrome and hypertensive crisis) [38]
Adding a TCA to an existing regimen (augmentation)
- Adding TCA to fluoxetine, paroxetine, or bupropion:
- Reduce TCA starting dose by ≥50% and use TDM guidance [46,58]
- CYP2D6 inhibition can increase TCA levels 200–800%
- Adding low-dose TCA for pain or insomnia (e.g., amitriptyline 10–25 mg):
- Interaction risk is lower at sub-antidepressant doses, but awareness of additive anticholinergic and serotonergic effects remains necessary
- Adding TCA to clomipramine or high-dose SSRI:
- Monitor closely for serotonin syndrome and QTc prolongation [38,60]
Switching between TCAs
- Direct switch (stop one TCA, start the next day) is generally feasible given shared class pharmacology [56]
- Tertiary to secondary amine switch (e.g., amitriptyline to nortriptyline):
- Start nortriptyline at approximately half the amitriptyline dose (nortriptyline is ~2× as potent on a mg basis); titrate with TDM guidance
- Switching to or from clomipramine:
- Cross-taper cautiously over 2–4 weeks due to its serotonergic profile [56,60]
- Monitor for serotonin syndrome during the overlap period
Brand names
Note: Most original branded TCA products in the US have been discontinued; TCAs are now predominantly dispensed as generics. Availability of specific branded formulations varies by country and may change over time.
Amitriptyline
- US: Elavil (discontinued), Endep (discontinued), Vanatrip (discontinued); Limbitrol (+ chlordiazepoxide; brand discontinued, generic available)
- Canada: APO-Amitriptyline, Bio-Amitriptyline, DOM-Amitriptyline, MINT-Amitriptyline, PMS-Amitriptyline
- Other countries/regions: Adepril, Adepress, Amineurin, Amirol, Amitop, Amitriptylin, Amitriptilina, Amitryp, Amytryptilinum, Amyzol, Anapsique, Conmitrip, Deprelio, Domical, Elatrol, Elatrolet, Endep, Fiorda, Kamitrin, Laroxyl, Latilin, Lentizol, Levate, Maxitrip, Mutabon (+ perphenazine), Novoprotect, Pertriptyl, Redomex, Saroten, Sarotex, Sarotex Retard, Syneudon, Trepiline, Triptyl, Tryptanol, Tryptizol, Uxen Retard
Nortriptyline
- US: Pamelor (discontinued), Aventyl (discontinued)
- Canada: APO-Nortriptyline, JAMP-Nortriptyline, MINT-Nortriptyline, PMS-Nortriptyline, TEVA-Nortriptyline
- Other countries/regions: Allegron, Ateben, Martimil, Motival (+ fluphenazine), Norpress, Nortrilen, Noritren, Nortimil, Nortriptylin, Nortriptilina, Pamelor, Paxtibi, Sensival, Vividyl
Clomipramine
- US: Anafranil
- Canada: APO-Clomipramine, CO Clomipramine, JAMP-Clomipramine, MYLAN-Clomipramine, PMS-Clomipramine
- Other countries/regions: Anafranil, Anafranil SR, Clofranil, Clomicalm, Clomipramina, Clopress, Equinorm, Gromin, Hydiphen, Klomipramin, Maronil, Placil
Imipramine
- US: Tofranil (imipramine HCl), Tofranil-PM (imipramine pamoate)
- Canada: Impril, PMS-Imipramine, TEVA-Imipramine
- Other countries/regions: Antideprin, Depresonil, Deprinol, Depsonil, Ethipramine, Imavate, Imidol, Imipramina, Irmin, Janimine, Melipramin, Melipramine, Praminil, Presamine, Primonil, Psychoforin, Sermonil, Surplix, Talpramin, Tipramine, Tofranil
Desipramine
- US: Norpramin
- Canada: Not widely available as a branded product
- Other countries/regions: Deprexan, Desipramin, Nebril, Norpramin, Pertofran, Pertofrane, Petylyl
Doxepin
- US: Silenor (3 mg, 6 mg — FDA-approved for insomnia); Sinequan (discontinued); Zonalon, Prudoxin (topical — for pruritus)
- Canada: APO-Doxepin, Sinequan
- Other countries/regions: Aponal, Doneurin, Doxal, Doxepia, Doxepin, Doxépine, Gilex, Ichderm (topical), Mareen, Quitaxon, Sinequan, Spectra, Xepin (topical)
Trimipramine
- US: Surmontil
- Canada: APO-Trimip, Rhotrimine, Surmontil
- Other countries/regions: Herphonal, Sapilent, Stangyl, Surmontil, Trimidura, Trimipramin, Trimineurin
Protriptyline
- US: Vivactil (discontinued)
- Canada: Not widely available
- Other countries/regions: Concordin, Vivactil
Combination products
- Amitriptyline + chlordiazepoxide: Limbitrol/Limbitrol DS (US brand discontinued; generic available) — indicated for depression with comorbid anxiety
- Amitriptyline + perphenazine: Mutabon, Etrafon, Triavil (various markets; most discontinued); indicated for depression with comorbid anxiety or agitation
- Nortriptyline + fluphenazine: Motival (some international markets)
References
1. Richelson, E. (1994). Pharmacology of Antidepressants—Characteristics of the Ideal Drug. Mayo Clinic Proceedings, 69(11), 1069–1081. https://doi.org/10.1016/S0025-6196(12)61375-5
2. Moraczewski, J., Awosika, A. O., & Aedma, K. K. (2023). Tricyclic Antidepressants. In StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/books/NBK557791/
3. Schatzberg, A. F., & Nemeroff, C. B. (Eds.). (2017). The American Psychiatric Association Publishing textbook of psychopharmacology (Fifth edition, DSM-5 edition). American Psychiatric Association Publishing.
4. DailyMed. (2021). Clomipramine hydrochloride capsules. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=0bf3c4f3-cf70-490d-89d8-9fb84962489e&type=pdf
5. Kurpius, M. P., & Alexander, B. (2006). Rates of in vivo methylation of desipramine and nortriptyline. Pharmacotherapy, 26(4), 505–510. https://doi.org/10.1592/phco.26.4.505
6. DailyMed. (2025). Amitriptyline hydrochloride tablets. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=33807ee6-8443-4613-8bbe-d66f6010fc60&type=pdf
7. DailyMed. (2023). Nortriptyline hydrochloride capsules. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=b1d9582c-742c-91a8-c2b3-bbdb21220470&type=pdf
8. Taylor, D., Poulou, S., & Clark, I. (2024). The cardiovascular safety of tricyclic antidepressants in overdose and in clinical use. Therapeutic Advances in Psychopharmacology, 14, 20451253241243297. https://doi.org/10.1177/20451253241243297
9. Cipriani, A., Furukawa, T. A., Salanti, G., Chaimani, A., Atkinson, L. Z., Ogawa, Y., Leucht, S., Ruhe, H. G., Turner, E. H., Higgins, J. P. T., Egger, M., Takeshima, N., Hayasaka, Y., Imai, H., Shinohara, K., Tajika, A., Ioannidis, J. P. A., & Geddes, J. R. (2018). Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: A systematic review and network meta-analysis. The Lancet, 391(10128), 1357–1366. https://doi.org/10.1016/S0140-6736(17)32802-7
10. Undurraga, J., & Baldessarini, R. J. (2017). Direct comparison of tricyclic and serotonin-reuptake inhibitor antidepressants in randomized head-to-head trials in acute major depression: Systematic review and meta-analysis. Journal of Psychopharmacology, 31(9), 1184–1189. https://doi.org/10.1177/0269881117711709
11. Undurraga, J., Vázquez, G. H., Tondo, L., & Baldessarini, R. J. (2020). Antidepressant responses in direct comparisons of melancholic and non-melancholic depression. Journal of Psychopharmacology, 34(12), 1335–1341. https://doi.org/10.1177/0269881120953983
12. Bandelow, B., Allgulander, C., Baldwin, D. S., Costa, D. L. da C., Denys, D., Dilbaz, N., Domschke, K., Hollander, E., Kasper, S., Möller, H.-J., Eriksson, E., Fineberg, N. A., Hättenschwiler, J., Kaiya, H., Karavaeva, T., Katzman, M. A., Kim, Y.-K., Inoue, T., Lim, L., … Zohar, J. (2023). World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for treatment of anxiety, obsessive-compulsive and posttraumatic stress disorders – Version 3. Part II: OCD and PTSD. World J. Biol. Psychiatry, 24(2), 118–134. https://doi.org/10.1080/15622975.2022.2086296
13. Swierkosz-Lenart, K., Dos Santos, J. F. A., Elowe, J., Clair, A.-H., Bally, J. F., Riquier, F., Bloch, J., Draganski, B., Clerc, M.-T., Pozuelo Moyano, B., Von Gunten, A., & Mallet, L. (2023). Therapies for obsessive-compulsive disorder: Current state of the art and perspectives for approaching treatment-resistant patients. Frontiers in Psychiatry, 14, 1065812. https://doi.org/10.3389/fpsyt.2023.1065812
14. Bandelow, B., Allgulander, C., Baldwin, D. S., Costa, D. L. da C., Denys, D., Dilbaz, N., Domschke, K., Eriksson, E., Fineberg, N. A., Hättenschwiler, J., Hollander, E., Kaiya, H., Karavaeva, T., Kasper, S., Katzman, M., Kim, Y.-K., Inoue, T., Lim, L., Masdrakis, V., … Zohar, J. (2023). World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for treatment of anxiety, obsessive-compulsive and posttraumatic stress disorders – Version 3. Part I: Anxiety disorders. World J. Biol. Psychiatry, 24(2), 79–117. https://doi.org/10.1080/15622975.2022.2086295
15. Skapinakis, P., Caldwell, D. M., Hollingworth, W., Bryden, P., Fineberg, N. A., Salkovskis, P., Welton, N. J., Baxter, H., Kessler, D., Churchill, R., & Lewis, G. (2021). Pharmacological and Psychotherapeutic Interventions for Management of Obsessive-compulsive Disorder in Adults: A Systematic Review and Network Meta-analysis. Focus, 19(4), 457–467. https://doi.org/10.1176/appi.focus.19402
16. Pallanti, S., & Quercioli, L. (2006). Treatment-refractory obsessive-compulsive disorder: Methodological issues, operational definitions and therapeutic lines. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 30(3), 400–412. https://doi.org/10.1016/j.pnpbp.2005.11.028
17. Finnerup, N. B., Attal, N., Haroutounian, S., McNicol, E., Baron, R., Dworkin, R. H., Gilron, I., Haanpää, M., Hansson, P., Jensen, T. S., Kamerman, P. R., Lund, K., Moore, A., Raja, S. N., Rice, A. S. C., Rowbotham, M., Sena, E., Siddall, P., Smith, B. H., & Wallace, M. (2015). Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. The Lancet. Neurology, 14(2), 162–173. https://doi.org/10.1016/S1474-4422(14)70251-0
18. Saarto, T., & Wiffen, P. J. (2007). Antidepressants for neuropathic pain. The Cochrane Database of Systematic Reviews, 2007(4), CD005454. https://doi.org/10.1002/14651858.CD005454.pub2
19. Moore, R. A., Derry, S., Aldington, D., Cole, P., & Wiffen, P. J. (2015). Amitriptyline for neuropathic pain in adults. The Cochrane Database of Systematic Reviews, 2015(7), CD008242. https://doi.org/10.1002/14651858.CD008242.pub3
20. Xu, X., Liu, Y., Dong, M., Zou, D., & Wei, Y. (2017). Tricyclic antidepressants for preventing migraine in adults. Medicine, 96(22), e6989. https://doi.org/10.1097/MD.0000000000006989
21. Jackson, J. L., Shimeall, W., Sessums, L., DeZee, K. J., Becher, D., Diemer, M., Berbano, E., & O’Malley, P. G. (2010). Tricyclic antidepressants and headaches: Systematic review and meta-analysis. BMJ, 341, c5222–c5222. https://doi.org/10.1136/bmj.c5222
22. Holroyd, K. A., O’Donnell, F. J., Stensland, M., Lipchik, G. L., Cordingley, G. E., & Carlson, B. W. (2001). Management of Chronic Tension-Type Headache With Tricyclic Antidepressant Medication, Stress Management Therapy, and Their Combination. JAMA : The Journal of the American Medical Association, 285(17), 2208–2215. https://doi.org/10.1001/jama.285.17.2208
23. Skaer, T. L. (2014). Fibromyalgia: Disease Synopsis, Medication Cost Effectiveness and Economic Burden. PharmacoEconomics, 32(5), 457–466. https://doi.org/10.1007/s40273-014-0137-y
24. Häuser, W., Wolfe, F., Tölle, T., Üçeyler, N., & Sommer, C. (2012). The Role of Antidepressants in the Management of Fibromyalgia Syndrome: A Systematic Review and Meta-Analysis. CNS Drugs, 26(4), 297–307. https://doi.org/10.2165/11598970-000000000-00000
25. Ford, A. C., Moayyedi, P., Chey, W. D., Harris, L. A., Lacy, B. E., Saito, Y. A., Quigley, E. M. M., & ACG Task Force on Management of Irritable Bowel Syndrome. (2018). American College of Gastroenterology Monograph on Management of Irritable Bowel Syndrome. The American Journal of Gastroenterology, 113, 1–18. https://doi.org/10.1038/s41395-018-0084-x
26. Talley, N. J., Locke, G. R., Saito, Y. A., Almazar, A. E., Bouras, E. P., Howden, C. W., Lacy, B. E., DiBaise, J. K., Prather, C. M., Abraham, B. P., El-Serag, H. B., Moayyedi, P., Herrick, L. M., Szarka, L. A., Camilleri, M., Hamilton, F. A., Schleck, C. D., Tilkes, K. E., & Zinsmeister, A. R. (2015). Effect of Amitriptyline and Escitalopram on Functional Dyspepsia: A Multi-Center, Randomized, Controlled Study. Gastroenterology, 149(2), 340–349.e2. https://doi.org/10.1053/j.gastro.2015.04.020
27. Ford, A. C., Wright-Hughes, A., Alderson, S. L., Ow, P.-L., Ridd, M. J., Foy, R., Bianco, G., Bishop, F. L., Chaddock, M., Cook, H., Cooper, D., Fernandez, C., Guthrie, E. A., Hartley, S., Herbert, A., Howdon, D., Muir, D. P., Nath, T., Newman, S., … Everitt, H. A. (2023). Amitriptyline at Low-Dose and Titrated for Irritable Bowel Syndrome as Second-Line Treatment in primary care (ATLANTIS): A randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet, 402(10414), 1773–1785. https://doi.org/10.1016/S0140-6736(23)01523-4
28. DailyMed. (2023). Silenor (doxepin hydrochloride) tablets. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=1bec1223-5239-4eb6-a9e8-62444106d2c0&type=pdf
29. Krystal, A. D., Lankford, A., Durrence, H. H., Ludington, E., Jochelson, P., Rogowski, R., & Roth, T. (2011). Efficacy and Safety of Doxepin 3 and 6 mg in a 35-day Sleep Laboratory Trial in Adults with Chronic Primary Insomnia. Sleep, 34(10), 1433–1442. https://doi.org/10.5665/SLEEP.1294
30. Yeung, W.-F., Chung, K.-F., Yung, K.-P., & Ng, T. H.-Y. (2015). Doxepin for insomnia: A systematic review of randomized placebo-controlled trials. Sleep Medicine Reviews, 19, 75–83. https://doi.org/10.1016/j.smrv.2014.06.001
31. Schneider, J., Patterson, M., & Jimenez, X. F. (2019). Beyond depression: Other uses for tricyclic antidepressants. Cleveland Clinic Journal of Medicine, 86(12), 807–814. https://doi.org/10.3949/ccjm.86a.19005
32. Kennedy, S. H., Lam, R. W., McIntyre, R. S., Tourjman, S. V., Bhat, V., Blier, P., Hasnain, M., Jollant, F., Levitt, A. J., MacQueen, G. M., McInerney, S. J., McIntosh, D., Milev, R. V., Müller, D. J., Parikh, S. V., Pearson, N. L., Ravindran, A. V., & Uher, R. (2016). Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive Disorder. Canadian Journal of Psychiatry. Revue Canadienne de Psychiatrie, 61(9), 540–560. https://doi.org/10.1177/0706743716659417
33. Stein, D. J., Ipser, J. C., Seedat, S., Sager, C., & Amos, T. (2006). Pharmacotherapy for post traumatic stress disorder (PTSD). The Cochrane Database of Systematic Reviews, 2006(1), CD002795. https://doi.org/10.1002/14651858.CD002795.pub2
34. DailyMed. (2024). Imipramine hydrochloride. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=a5f60cf1-6f14-40a3-a794-9d69b28088d8
35. Caldwell, P. H., Sureshkumar, P., & Wong, W. C. (2016). Tricyclic and related drugs for nocturnal enuresis in children. The Cochrane Database of Systematic Reviews, 2016(1), CD002117. https://doi.org/10.1002/14651858.CD002117.pub2
36. Hughes, J. R., Stead, L. F., Hartmann-Boyce, J., Cahill, K., & Lancaster, T. (2014). Antidepressants for smoking cessation. The Cochrane Database of Systematic Reviews, 2014(1), CD000031. https://doi.org/10.1002/14651858.CD000031.pub4
37. Prochazka, A. V., Weaver, M. J., Keller, R. T., Fryer, G. E., Licari, P. A., & Lofaso, D. (1998). A Randomized Trial of Nortriptyline for Smoking Cessation. Archives of Internal Medicine, 158(18), 2035–2039. https://doi.org/10.1001/archinte.158.18.2035
38. Gillman, P. K. (2007). Tricyclic antidepressant pharmacology and therapeutic drug interactions updated. British Journal of Pharmacology, 151(6), 737–748. https://doi.org/10.1038/sj.bjp.0707253
39. Flint, A. J. (1997). Pharmacologic treatment of depression in late life. CMAJ: Canadian Medical Association Journal, 157(8), 1061–1067. https://pmc.ncbi.nlm.nih.gov/articles/PMC1228262/
40. Wisner, K. L., Perel, J. M., Findling, R. L., & Hinnes, R. L. (1997). Nortriptyline and its hydroxymetabolites in breastfeeding mothers and newborns. Psychopharmacology Bulletin, 33(2), 249–251. https://www.ncbi.nlm.nih.gov/pubmed/9230638
41. Tatsumi, M., Groshan, K., Blakely, R. D., & Richelson, E. (1997). Pharmacological profile of antidepressants and related compounds at human monoamine transporters. European Journal of Pharmacology, 340(2–3), 249–258. https://doi.org/10.1016/S0014-2999(97)01393-9
42. Roose, S. P., Glassman, A. H., Siris, S. G., Walsh, B. T., Bruno, R. L., & Wright, L. B. (1981). Comparison of imipramine- and nortriptyline-induced orthostatic hypotension: A meaningful difference. Journal of Clinical Psychopharmacology, 1(5), 316–319. https://doi.org/10.1097/00004714-198109000-00008
43. Thayssen, P., Bjerre, M., Kragh-Sørensen, P., Møller, M., Petersen, O. L., Kristensen, C. B., & Gram, L. F. (1981). Cardiovascular effects of imipramine and nortriptyline in elderly patients. Psychopharmacology, 74(4), 360–364. https://doi.org/10.1007/BF00432748
44. Hiemke, C., Bergemann, N., Clement, H., Conca, A., Deckert, J., Domschke, K., Eckermann, G., Egberts, K., Gerlach, M., Greiner, C., Gründer, G., Haen, E., Havemann-Reinecke, U., Hefner, G., Helmer, R., Janssen, G., Jaquenoud, E., Laux, G., Messer, T., … Baumann, P. (2018). Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology: Update 2017. Pharmacopsychiatry, 51(01/02), 9–62. https://doi.org/10.1055/s-0043-116492
45. Ericksen, S. E. (1979). Recent advances in antidepressant drug treatment. The Western Journal of Medicine, 131(2), 104–113. https://www.ncbi.nlm.nih.gov/pubmed/390888
46. Hicks, J., Sangkuhl, K., Swen, J., Ellingrod, V., Müller, D., Shimoda, K., Bishop, J., Kharasch, E., Skaar, T., Gaedigk, A., Dunnenberger, H., Klein, T., Caudle, K., & Stingl, J. (2017). Clinical pharmacogenetics implementation consortium guideline (CPIC) for CYP2D6 and CYP2C19 genotypes and dosing of tricyclic antidepressants: 2016 update. Clinical Pharmacology & Therapeutics, 102(1), 37–44. https://doi.org/10.1002/cpt.597
47. Zhou, S., Li, P., Lyu, X., Lai, X., Liu, Z., Zhou, J., Liu, F., Tao, Y., Zhang, M., Yu, X., Tian, J., & Sun, F. (2025). Efficacy and dose–response relationships of antidepressants in the acute treatment of major depressive disorders: A systematic review and network meta-analysis. Chinese Medical Journal, 138(12), 1433–1438. https://doi.org/10.1097/CM9.0000000000003138
48. Baethge, C., Braun, C., Rink, L., Schwarzer, G., Henssler, J., & Bschor, T. (2022). Dose effects of tricyclic antidepressants in the treatment of acute depression – A systematic review and meta-analysis of randomized trials. Journal of Affective Disorders, 307, 191–198. https://doi.org/10.1016/j.jad.2022.03.075
49. Biederman, J. (1991). Sudden death in children treated with a tricyclic antidepressant. Journal of the American Academy of Child and Adolescent Psychiatry, 30(3), 495–498. https://doi.org/10.1097/00004583-199105000-00023
50. Riddle, M. A., Geller, B., & Ryan, N. (1993). Another sudden death in a child treated with desipramine. Journal of the American Academy of Child and Adolescent Psychiatry, 32(4), 792–797. https://doi.org/10.1097/00004583-199307000-00013
51. Riemann, D., Voderholzer, U., Cohrs, S., Rodenbeck, A., Hajak, G., Rüther, E., Wiegand, M. H., Laakmann, G., Baghai, T., Fischer, W., Hoffmann, M., Hohagen, F., Mayer, G., & Berger, M. (2002). Trimipramine in primary insomnia: Results of a polysomnographic double-blind controlled study. Pharmacopsychiatry, 35(5), 165–174. https://doi.org/10.1055/s-2002-34119
52. DailyMed. (2026). Trimipramine capsule. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=4f31df66-7dc2-1f04-e054-00144ff88e88&type=pdf
53. DailyMed. (2023). Protriptyline hydrochloride tablets. https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=c6c7c39e-e510-4f4a-bf7a-926f0e075d3a&type=pdf
54. Moody, J. P., Whyte, S. F., MacDonald, A. J., & Naylor, G. J. (1977). Pharmacokinetic aspects of protriptyline plasma levels. European Journal of Clinical Pharmacology, 11(1), 51–56. https://doi.org/10.1007/BF00561788
55. Labbate, L. A. (Ed.). (2010). Handbook of psychiatric drug therapy (6th ed). Wolters Kluwer Health/Lippincott Williams & Wilkins.
56. Keks, N., Hope, J., & Keogh, S. (2016). Switching and stopping antidepressants. Australian Prescriber, 39(3), 76–83. https://doi.org/10.18773/austprescr.2016.039
57. Ogle, N. R., & Akkerman, S. R. (2013). Guidance for the Discontinuation or Switching of Antidepressant Therapies in Adults. Journal of Pharmacy Practice, 26(4), 389–396. https://doi.org/10.1177/0897190012467210
58. U.S. Food and Drug Administration. (2023). Prozac. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/018936%20s112lbl.pdf
59. Juřica, J., & Žourková, A. (2013). Dynamics and persistence of CYP2D6 inhibition by paroxetine. Journal of Clinical Pharmacy and Therapeutics, 38(4), 294–300. https://doi.org/10.1111/jcpt.12042
60. Garel, N., Greenway, K. T., Tabbane, K., & Joober, R. (2021). Serotonin syndrome: SSRIs are not the only culprit. Journal of Psychiatry & Neuroscience : JPN, 46(3), E369–E370. https://doi.org/10.1503/jpn.210001



