In a nutshell
Gastrointestinal (GI) side effects are the most common adverse effects of antidepressants and the leading cause of early treatment discontinuation. Nausea affects roughly 25% of patients on SSRIs/SNRIs. Duloxetine carries the highest nausea risk of any commonly used antidepressant; vilazodone carries the highest diarrhea risk; and mirtazapine is one of the best-tolerated agents for the GI tract.
- GI profiles at a glance:
- Best overall GI tolerability:
- Mirtazapine (no significant nausea, diarrhea, or anorexia, but increases appetite and weight)
- Bupropion (minimal serotonergic GI effects; low nausea, no significant diarrhea)
- Agomelatine (bypasses serotonergic GI pathways; no significant nausea or dry mouth)
- Highest nausea risk: Duloxetine, levomilnacipran, vilazodone
- Nausea-sparing alternatives: mirtazapine, trazodone, agomelatine, bupropion
- Highest diarrhea risk: Vilazodone, sertraline
- Most SNRIs, TCAs, and mirtazapine do not significantly increase diarrhea
- Highest constipation risk: Amitriptyline, clomipramine, reboxetine
- Most SSRIs (except paroxetine) are constipation-sparing
- Highest dry mouth risk: Amitriptyline, reboxetine, trazodone
- Vortioxetine and agomelatine show no significant dry mouth
- Best overall GI tolerability:

- Key clinical recommendations:
- Agent selection by patient profile:
- In nausea-sensitive patients:
- Prefer mirtazapine, agomelatine, or bupropion
- Avoid duloxetine, venlafaxine, vilazodone
- In IBS-D or diarrhea-prone patients:
- Prefer low-dose amitriptyline or mirtazapine
- Avoid sertraline, vilazodone, fluvoxamine
- In IBS-C or elderly patients with anticholinergic burden:
- Prefer fluoxetine, sertraline, or escitalopram
- Avoid amitriptyline, clomipramine, reboxetine, paroxetine
- In nausea-sensitive patients:
- Start low, go slow: Initiate at the lowest effective dose and titrate gradually to reduce peak serotonergic stimulation in the gut
- Educate early and set realistic expectations: Counsel patients that serotonergic nausea typically resolves within 1–2 weeks through 5-HT3 receptor desensitization; providing specific timelines and positive framing reduces nocebo-driven symptoms and premature discontinuation.
- Consider switching if GI symptoms persist: If GI symptoms persist beyond 3–4 weeks despite dose optimization and supportive measures, switch to a better-tolerated agent rather than waiting for spontaneous resolution.
- Agent selection by patient profile:
- Symptom-specific management essentials:
- Nausea:
- Take with food, ginger (~1 g/day), evening dosing
- If symptoms persist and switching is not an option: ondansetron 4 mg PRN or low-dose mirtazapine augmentation
- Diarrhea:
- Dietary modifications (avoid caffeine/lactose), loperamide PRN
- Consider switching from sertraline/vilazodone to a lower-risk agent
- Constipation:
- First-line: increase fiber, fluids, and activity
- If non-pharmacological measures are insufficient: polyethylene glycol (PEG) 17 g daily; consider switching from TCAs to SSRIs
- Dry mouth:
- Frequent sips of water, sugar-free gum, saliva substitutes
- Fluoride rinses for dental protection
- Pilocarpine for severe refractory cases
- Nausea:
Introduction
- GI side effects appear early, typically during the first few weeks of treatment, when the risk of patient nonadherence is highest [1]
- SSRIs are more commonly associated with nausea, decreased appetite, and diarrhea; TCAs are more prone to constipation, dry mouth, and weight gain due to anticholinergic effects [2–4]
- Among SSRIs, sertraline shows a higher incidence of diarrhea compared to other SSRIs
- Among SNRIs, venlafaxine is associated with higher rates of nausea and vomiting compared to duloxetine
- The high nocebo rate (71% adverse event incidence in placebo groups) underscores the importance of setting realistic expectations and providing specific timelines for side effect resolution [5]
- Clinicians should inform patients about both the frequency of common gastrointestinal events and the substantial contribution of expectation-related symptoms.
- Emotional alterations may cause intestinal disturbances and vice versa through the microbiome-gut-brain axis [6,7]
- Patients with MDD frequently present with gastrointestinal somatic symptoms as part of their depressive presentation [2]
Mechanisms of Antidepressant-Induced GI Side Effects
The Gut–Brain Axis and Peripheral Serotonin
- The gut contains most of the body’s serotonin (5-HT), produced predominantly by enterochromaffin (EC) cells; serotonergic signaling is essential for physiological gastrointestinal motility [8,9]
- The serotonin transporter (SERT) is expressed in the GI tract as well as the CNS [1,2]
- Drugs that inhibit SERT therefore raise serotonin availability in both compartments, making GI effects among the most predictable pharmacological consequences of serotonergic antidepressants.

Serotonergic Pathways
- Serotonergic modulation is the dominant mechanism underlying antidepressant-induced nausea/vomiting, diarrhea, and abdominal pain [2,3,10]
- Drugs that affect serotonin levels or serotonin receptors demonstrate the highest incidence of nausea and vomiting [3]
Nausea and vomiting: 5-HT3 receptor–mediated emesis
- The 5-HT3 receptor is unique among serotonin receptors as it is a ligand-gated ion channel, mediating rapid synaptic transmission [11]

- These receptors are densely populated in two critical locations:
- The Chemoreceptor Trigger Zone (CTZ) of the area postrema in the brainstem, which lies outside the blood-brain barrier and is chemically accessible to the circulation
- Vagal afferent terminals lining the gastrointestinal mucosa
- Elevated 5-HT stimulates 5-HT3 receptors on vagal afferents, triggering signals to brainstem emetic centers which initiate the nausea and vomiting response [1,11]
- Clinical implications:
- Nausea tolerance:
- The initial nausea caused by SSRIs typically resolves within 7–14 days.
- Chronic stimulation of 5-HT3 receptors leads to desensitization; the receptors effectively “turn down the volume” on the emetic signal [12]
- Mirtazapine as an antiemetic:
- Mirtazapine is a 5-HT3 antagonist — this explains its association with significantly lower nausea rates
- It is used off-label as an antiemetic in palliative care and hyperemesis gravidarum [13–15]
- Ondansetron and 5-HT3 antagonists in clinical practice:
- Ondansetron exploits this same receptor pathway and is standard of care for chemotherapy-induced nausea
- It can be used at 4–8 mg PRN to counteract antidepressant-induced nausea directly
- Nausea tolerance:
Motility and Diarrhea: 5-HT4 Receptor
- SERT inhibition in the gut increases local 5-HT availability, accelerating peristalsis and increasing fluid secretion into the intestinal lumen [1]
- The 5-HT4 receptor enhances the release of acetylcholine and promotes colonic motility [16,17]
- This manifests clinically as diarrhea or loose stools, a side effect particularly notable with agents like sertraline and vilazodone.
- Clinical implications:
- The pro-kinetic effect of 5-HT4 stimulation can be harnessed therapeutically:
- SSRIs are often the preferred antidepressant in patients with constipation-predominant irritable bowel syndrome (IBS-C), where the GI side effect becomes the treatment [18,19]
Anticholinergic Pathways
- Muscarinic receptor blockade is the primary mechanism underlying antidepressant-induced constipation and contributes to dry mouth [2,3,20]
- Tricyclic antidepressants (TCAs) and paroxetine competitively block muscarinic acetylcholine receptors (specifically M2 and M3 subtypes) in the gut, which [21,22]
- Inhibits smooth muscle contraction
- Prolongs intestinal transit time
- Reduces mucosal secretions
Constipation
- TCAs carry the strongest anticholinergic burden and show the highest constipation incidence in a network meta-analysis [3]
- The majority of antidepressants showed higher constipation rates versus placebo, with the notable exception of most SSRIs, consistent with the lower peripheral anticholinergic activity of this class [3]
- This dissociation (TCAs driving constipation versus SSRIs driving diarrhea) reflects the contrasting influence of anticholinergic versus serotonergic pathways on gut motility [3,20]
Dry mouth
- Parasympathetic stimulation drives salivary secretion through cholinergic transmission at M3 muscarinic receptors
- Anticholinergic agents like amitriptyline and paroxetine block this pathway, reducing salivary flow
- SSRIs produce dry mouth by altering the composition of salivary proteins rather than directly suppressing secretion volume [2,23]
Noradrenergic Pathways
- Norepinephrine tends to slow gastric emptying and intestinal transit (the “fight or flight” response diverts blood from the gut and reduces digestive activity), which can contribute to constipation.
- Norepinephrine can also have an appetite-suppressing effect initially (some patients on SNRIs report reduced appetite or weight loss early on).
- Norepinephrine accumulation in the brainstem may also contribute to the higher dry mouth rates seen with SNRIs compared to SSRIs, through inhibition of parasympathetic salivary neurons [2]
Histaminergic Pathways
- H1 receptor antagonism—along with 5-HT3 and 5-HT2A antagonism—contributes to the protective effect against nausea and vomiting observed with agents like mirtazapine, trazodone, and amitriptyline [2,3,24]
- However, H1 antagonism simultaneously stimulates appetite and promotes weight gain [2,25]
Dose–Response Relationships

- Gastrointestinal effects do not always increase linearly with dose[3]
- Linear dose-dependent increase: Citalopram, fluoxetine, levomilnacipran, and venlafaxine show dose-dependent increases in nausea/vomiting; all agents with significant dry mouth demonstrate dose-dependent increases within conventional therapeutic ranges [3]
- Inverted U-shaped curve: Duloxetine and vortioxetine show nausea/vomiting peaking at mid-range doses before decreasing at higher doses; fluoxetine shows an inverted U-shape for diarrhea [3]
- Flat or decreasing curves: Escitalopram, paroxetine, and sertraline show a slight decrease in nausea risk at higher doses [3]
- Clinical implications:
- Dose adjustment may selectively mitigate specific gastrointestinal symptoms
- The optimal dose for tolerability does not always follow the “lower is always better” assumption [3]
Comparing GI Side Effect Risk Across Antidepressants
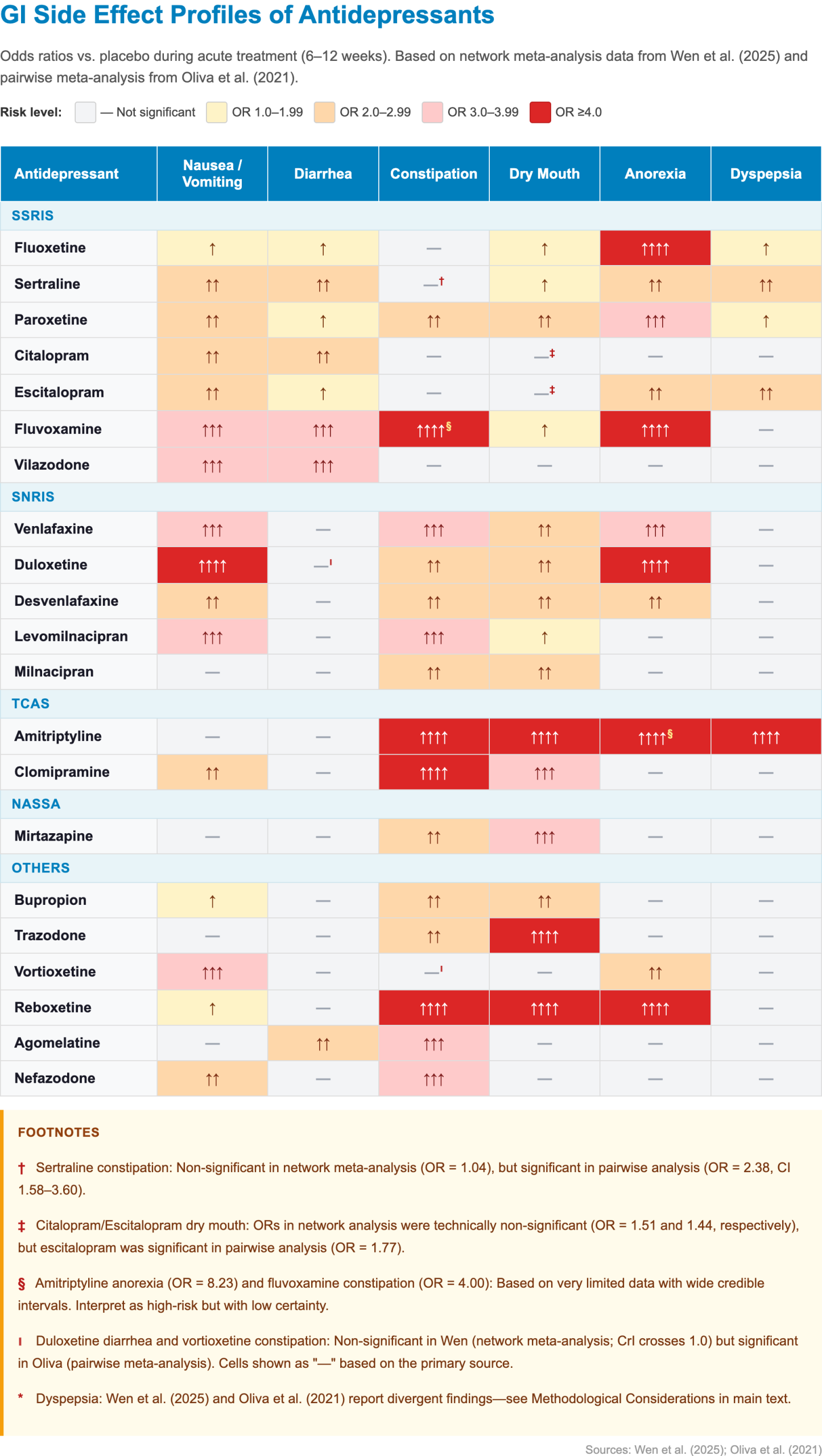
Nausea and Vomiting
- Nausea and vomiting are the most common GI adverse effects of antidepressant treatment and the most frequent cause of early discontinuation [3,26]
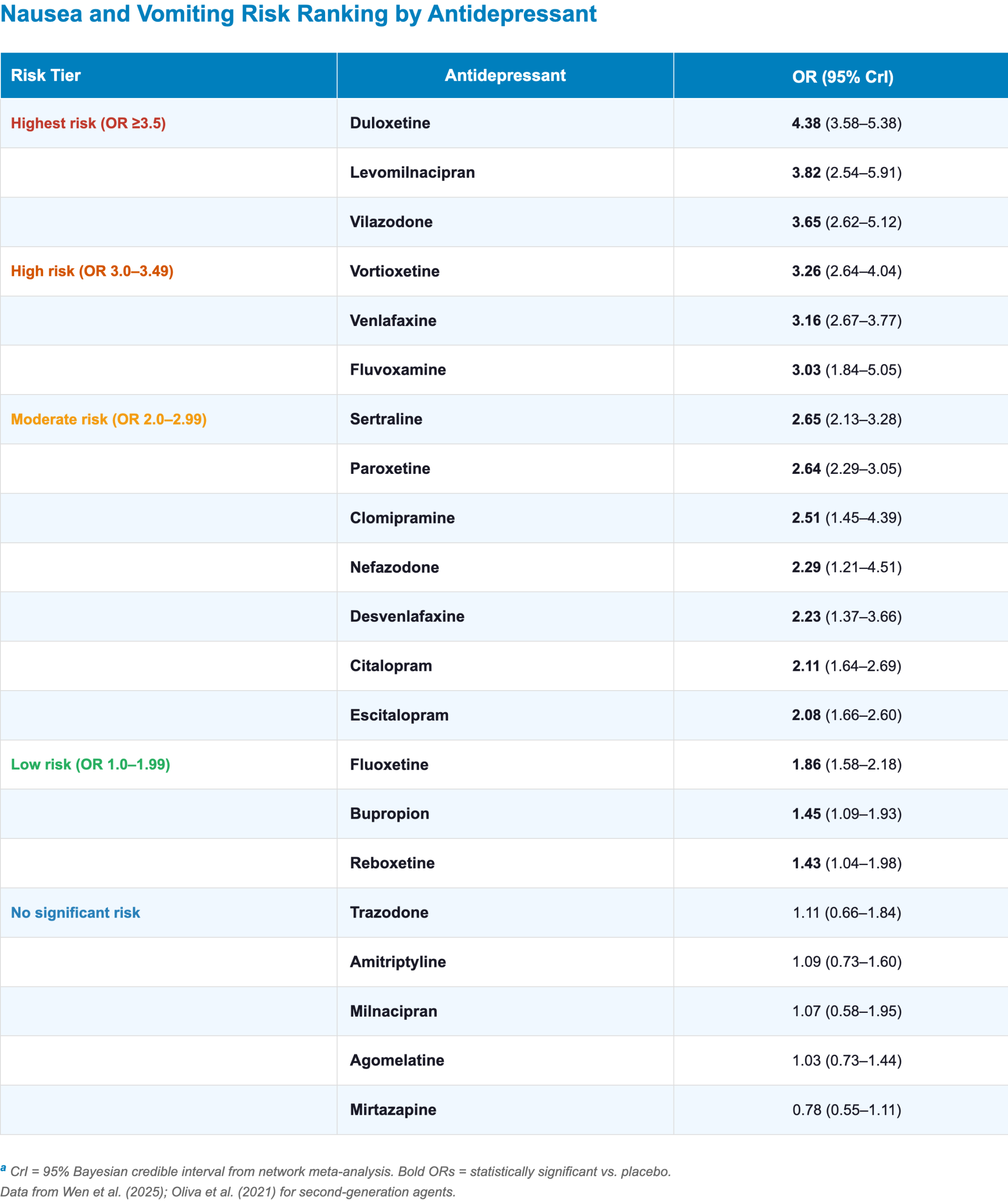
- Clinical highlights:
- Duloxetine: carries the highest nausea risk of any commonly used antidepressant [2,3]
- Vortioxetine: Nausea is dose-dependent—risk rises steeply from 5 mg to 10 mg, then plateaus or decreases at higher doses (inverted U-curve) [3]
- Starting at 5 mg and titrating slowly substantially improves early tolerability
- SSRIs:
- Fluoxetine carries the lowest nausea risk among SSRIs
- Sertraline and paroxetine show higher risk [3]
- Paroxetine and venlafaxine are less well tolerated than sertraline and escitalopram for overall adverse events across anxiety and depressive disorders [5]
- Mirtazapine: Shows no significant nausea association and may have active antiemetic properties via 5-HT3 antagonism [2,3]
- Trazodone, amitriptyline, and agomelatine: show no significant nausea vs. placebo, likely due to antihistaminergic (H1) and 5-HT2A antagonist properties [3]
- Dosing tips:
- For agents with linear dose–response (citalopram, fluoxetine, levomilnacipran, venlafaxine), slow titration is key — higher doses produce proportionally more nausea [3]
- For agents with flat curves (escitalopram, paroxetine, sertraline), nausea is present even at low doses and does not escalate markedly — tolerance development rather than dose adjustment is the strategy
Diarrhea
- Antidepressants that cause diarrhea generally do not also cause constipation (and vice versa), reflecting distinct mechanistic profiles [2]
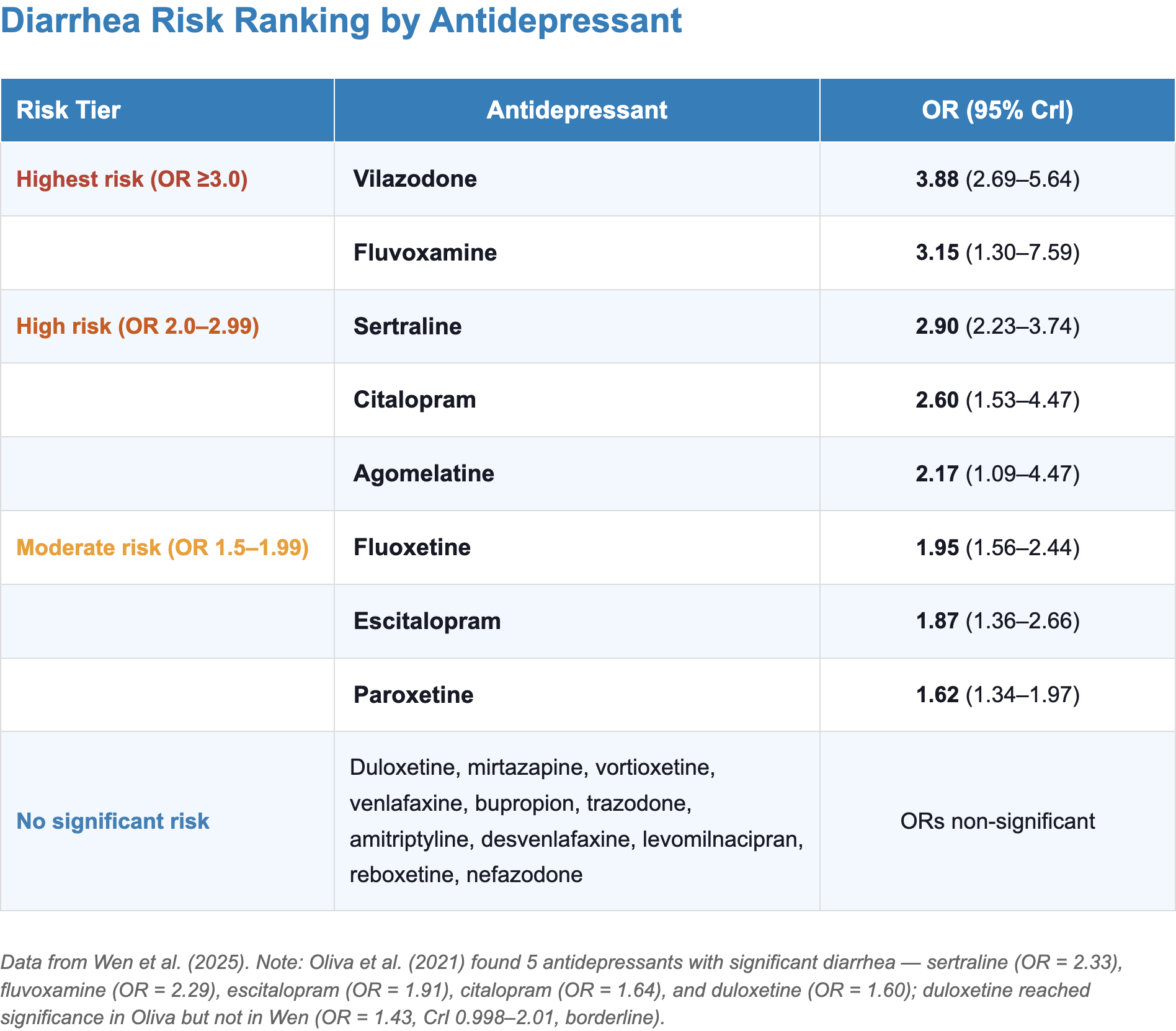
- Clinical highlights:
- Sertraline: consistently the highest-diarrhea SSRI across two major meta-analyses [2,3]
- Vilazodone: carries the highest absolute diarrhea risk of any agent (OR = 3.88) [3]
- Its dual SSRI + 5-HT1A partial agonist mechanism amplifies enteric serotonergic stimulation
- Diarrhea risk increases in a dose-dependent manner for escitalopram, fluoxetine, paroxetine, and sertraline [3]
- In IBS-D:
- Avoid: sertraline, vilazodone, fluvoxamine
- Prefer: low-dose amitriptyline, mirtazapine, or an agent with constipating properties
- In IBS-C: sertraline may serve a dual therapeutic purpose
Constipation
- Constipation is the dominant GI effect of anticholinergic antidepressants (TCAs, paroxetine) and agents with noradrenergic reuptake inhibition (SNRIs, reboxetine) [3]
- Unlike serotonergic nausea, anticholinergic constipation may not attenuate substantially with continued treatment, though systematic long-term data are limited [2]
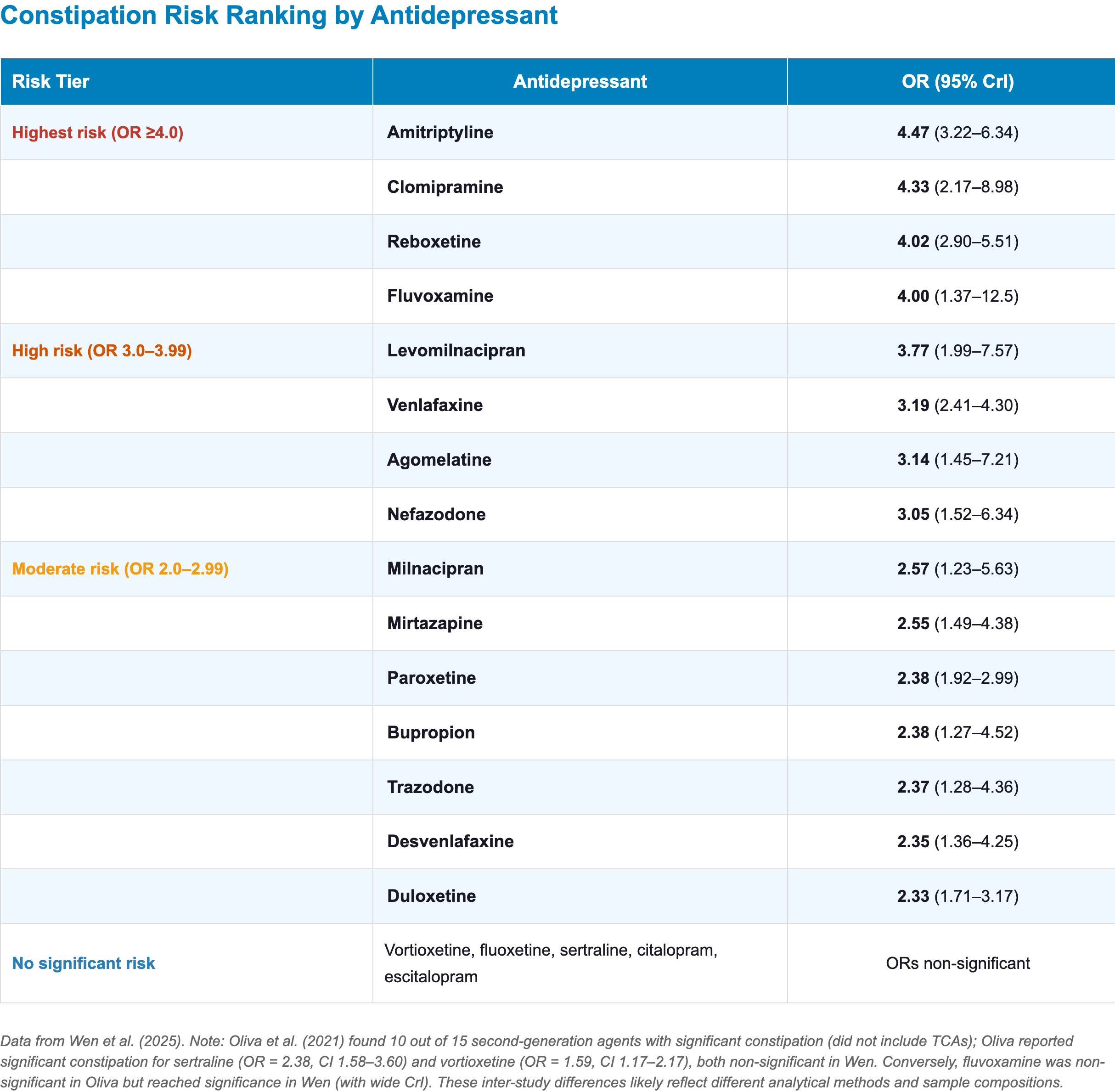
- Clinical highlights:
- TCAs: carry the highest constipation risk, driven by potent muscarinic receptor blockade that slows colonic transit [3,27]
- SNRIs: as a class carry a meaningful constipation risk, likely through norepinephrine-mediated reduction of colonic motility [3]
- SSRIs:
- Paroxetine: the only SSRI with consistently significant constipation across two major meta-analyses, reflecting its higher anticholinergic burden within the class [2,3]
- Constipation risk increases with dose [3]
- Fluoxetine, citalopram, and escitalopram: did not significantly increase constipation risk in two major meta-analyses — preferred options when constipation is a concern [2,3]
- Sertraline: mixed evidence — may carry some constipation risk, particularly at higher doses; monitor and consider alternatives if symptoms emerge [2,3]
- Paroxetine: the only SSRI with consistently significant constipation across two major meta-analyses, reflecting its higher anticholinergic burden within the class [2,3]
Dry Mouth
- Unlike serotonergic nausea, dry mouth does not predictably attenuate with continued use
- Long-term sequelae include dental caries, oral infections, and reduced quality of life [28]
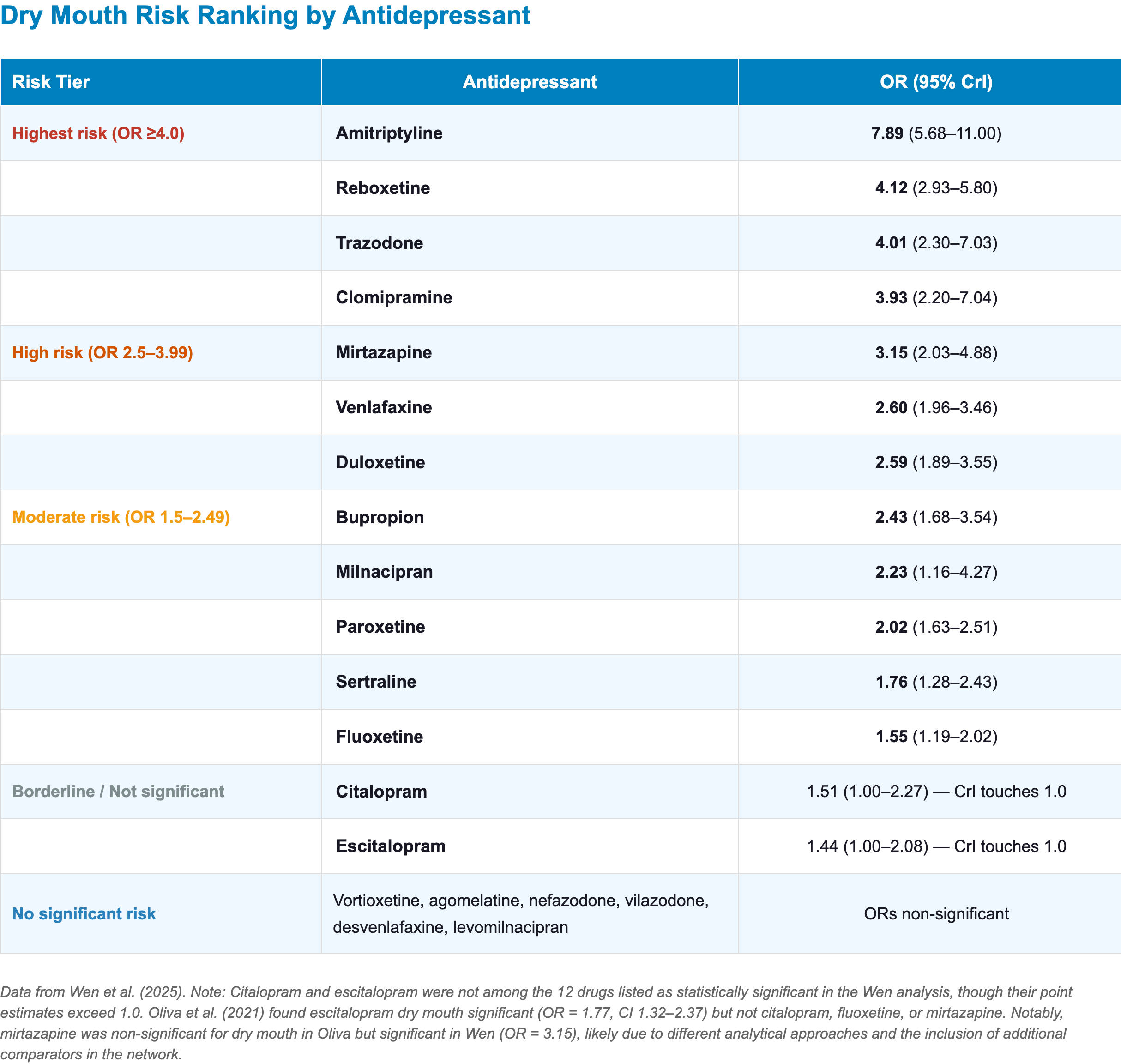
- Clinical highlights:
- Amitriptyline: carries the highest dry mouth risk—nearly 8× the odds vs. placebo—consistent with its potent muscarinic receptor binding affinity [3,28]
- Trazodone: shows unexpectedly high dry mouth despite minimal anticholinergic activity
- Likely mediated through α1-adrenergic blockade reducing parasympathetic outflow to salivary glands [3]
- Vortioxetine and agomelatine: show no significant dry mouth in either meta-analysis, making them favorable options when xerostomia is a concern [2,3]
Anorexia / Decreased Appetite
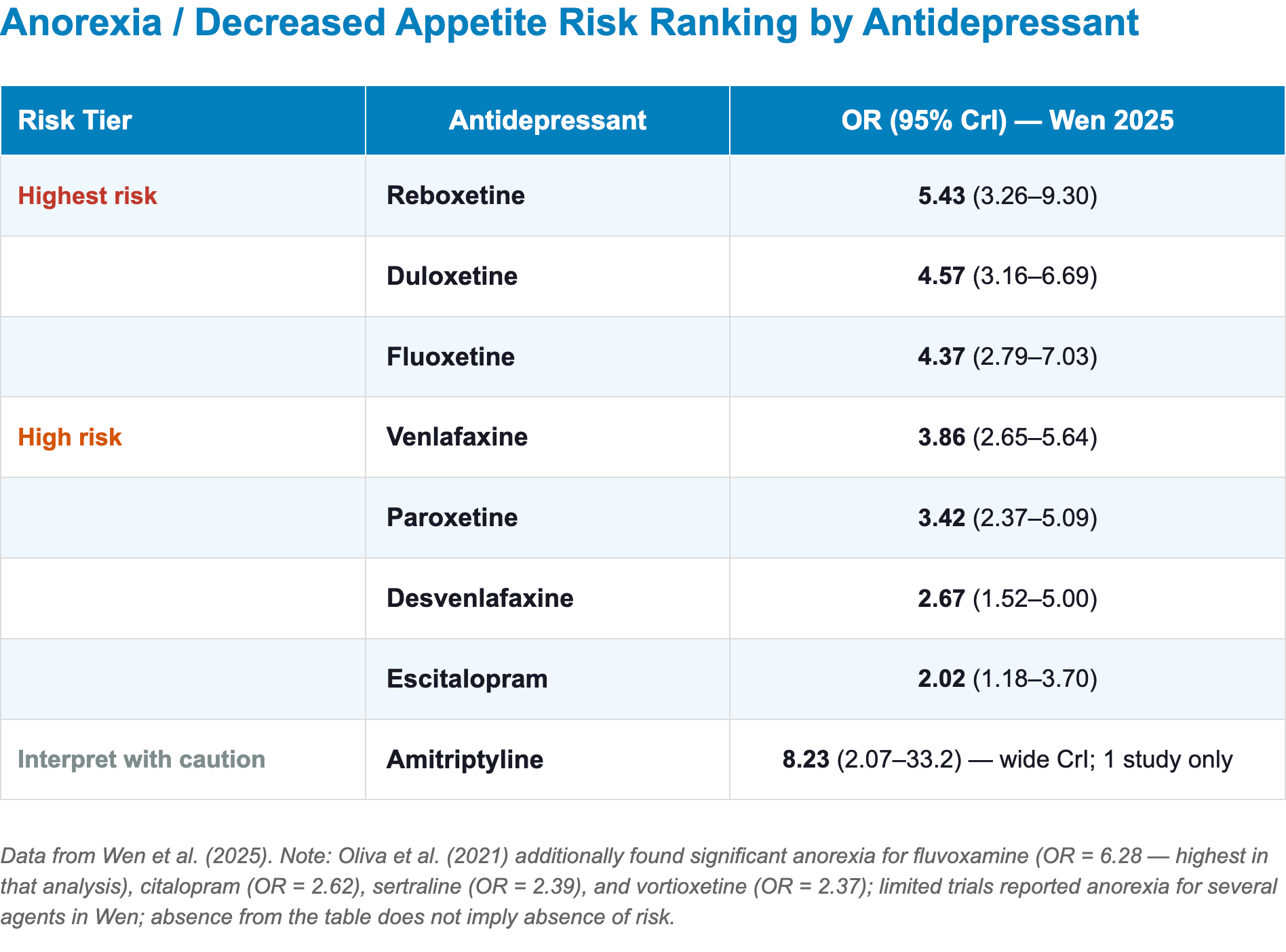
- Clinical highlights:
- Fluoxetine: one of the highest anorexia risks [25]
- The effect is biphasic — acute appetite suppression may be followed by gradual reversal over months
- Fluvoxamine: carries the highest anorexia risk among SSRIs with available data [2]
- Duloxetine and venlafaxine: combine serotonergic anorexia with noradrenergic-mediated appetite suppression, explaining their particularly high risk [3]
- Fluoxetine: one of the highest anorexia risks [25]
Dyspepsia
- Dyspepsia data are limited; only a few antidepressants (amitriptyline, fluoxetine, paroxetine, sertraline, escitalopram) show significantly higher dyspepsia vs. placebo across available evidence [2,3]
- Paradoxically, some antidepressants (particularly low-dose TCAs and SSRIs) are used therapeutically for functional dyspepsia [29,30]
- Amitriptyline 50 mg/day improved functional dyspepsia symptoms, while escitalopram did not differ from placebo in a multicenter RCT [29]
Management and Treatment Strategies
- Concerns about potential long-term side effects are the most frequent cause of spontaneous antidepressant discontinuation [2,3]
Choosing an Antidepressant Based on GI Risk
- Among the most actionable preventive strategies is selecting agents with a more favorable GI profile for the patient’s risk factors and prior tolerability
- Use the GI risk heatmap and clinical decision framework (see Comparing GI Profiles section) to guide initial agent selection
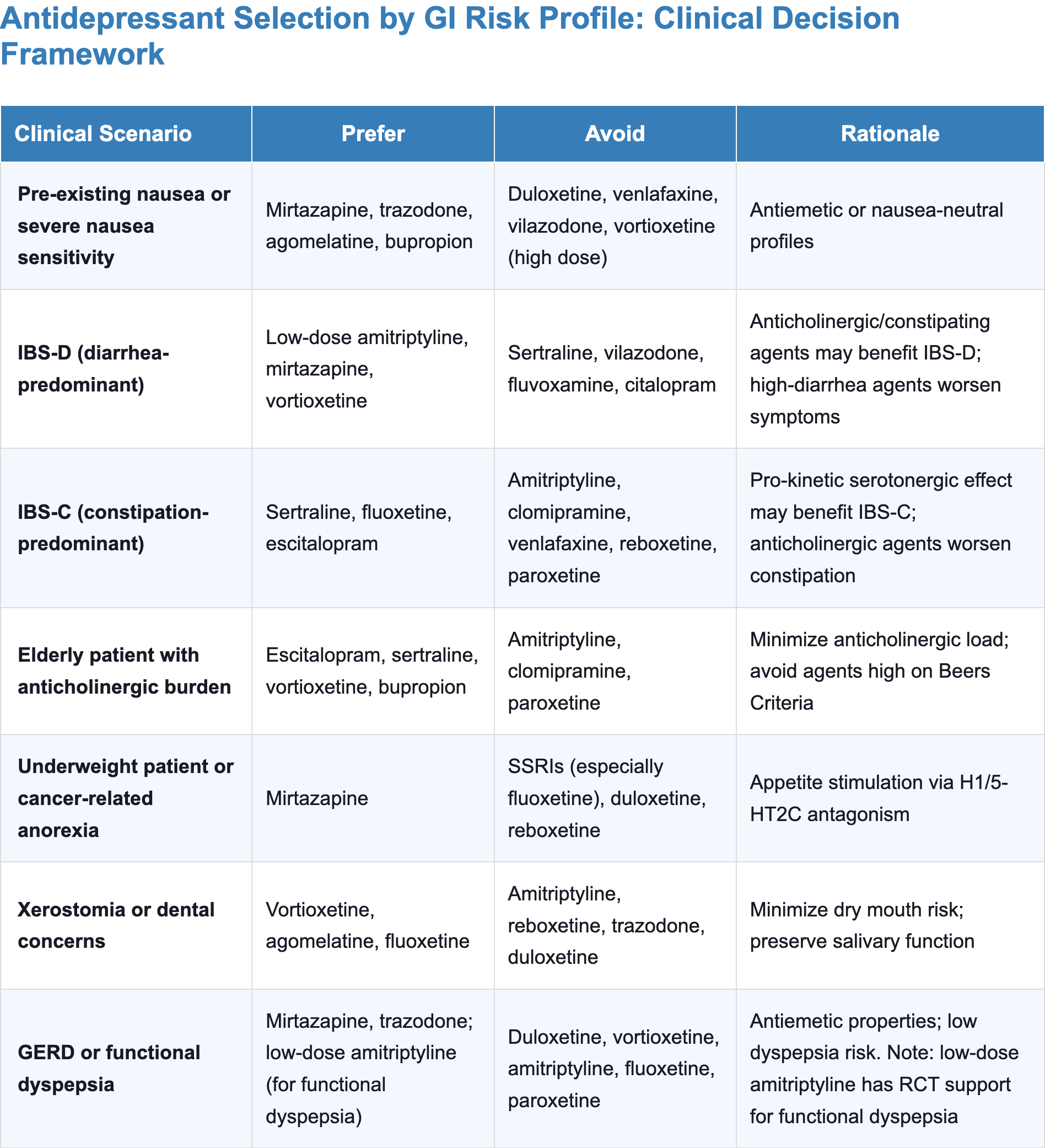
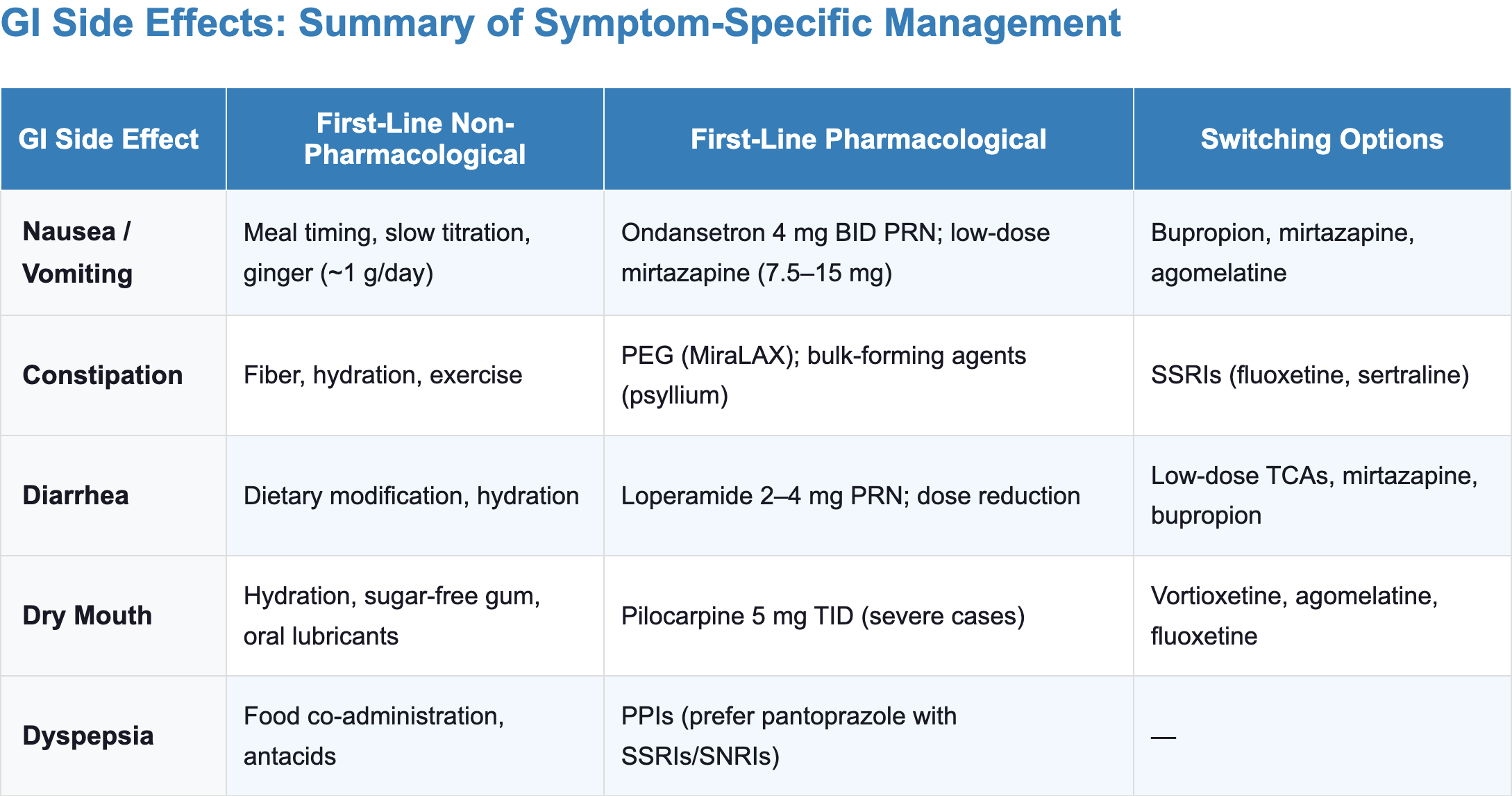
General principles
- Anticipate and educate:
- Setting expectations reduces distress, increases tolerance, and decreases premature discontinuation.
- Provide specific timelines: “Most patients find that nausea improves significantly by the end of the second week”
- A realistic, positive framing of potential side effects (rather than exhaustive warnings) may reduce nocebo-driven symptoms and improve adherence [5]
- Start low, go slow:
- Initiate at the lowest effective dose and titrate gradually [12,31]
- Slow titration reduces peak serotonergic stimulation in the GI tract which may reduce the incidence of early nausea and diarrhea [31]
- Clinical tip: Always consider starting at half the intended dose for SSRIs and SNRIs
- Take with food:
- Administering the antidepressant with a meal can reduce nausea, particularly for SSRIs and SNRIs
- If the patient typically skips breakfast, recommend moving the dose to a later meal time rather than taking on an empty stomach; even a small snack (crackers, toast, or a small amount of a fatty food such as peanut butter) can buffer gastric irritation
- This is mandatory for vilazodone (bioavailability is significantly reduced without food)
- Evening dosing:
- For agents causing nausea, evening administration allows the patient to sleep through the peak nausea period
- Paroxetine at night can let the patient sleep through any mild nausea.
- Mirtazapine is usually given at bedtime due to sedation, which also reduces patients’ awareness of its appetite stimulation
- Particularly useful for: venlafaxine, duloxetine, vortioxetine
- Note: Some agents (e.g., fluoxetine, bupropion) may cause insomnia and are better taken in the morning
- For agents causing nausea, evening administration allows the patient to sleep through the peak nausea period
- Consider formulation and peak-level effects:
- Extended-release formulations (venlafaxine XR, duloxetine delayed-release) may reduce peak-related GI symptoms compared to immediate-release forms [32,33]
- However, a systematic review did not find significant differences in the rate of adverse events (including nausea) between immediate versus extended-release venlafaxine [34]
- Clinical tips:
- Splitting the dose (even when pharmacokinetics do not require it) can reduce peak serotonin effects on the GI tract.
- If a patient takes two emetogenic medications, advise taking them after different meals rather than together
- For patients with bowel resections or short bowel, extended-release might not fully absorb [35]
- Use of temporary dose reduction and dose-dependent effects:
- If GI symptoms emerge at a higher dose, reducing to the previously tolerated dose may resolve symptoms while maintaining therapeutic effect (See Comparing GI Side effects, dose response patterns) [2,3]
- E.g., if a patient on sertraline 100 mg increases to 150 mg and develops vomiting, reduce back to 100 mg for another 1–2 weeks before reattempting titration
Symptomatic Management by GI Effect
Nausea and vomiting

- Non-pharmacological measures
- Watchful waiting: Reassure patients that nausea typically attenuates within 1–2 weeks through 5-HT3 receptor desensitization
- Take medication after food, such as crackers, toast, or peanut butter [12]
- Ginger (500–1000 mg, capsule or grated in tea) has evidence for antiemetic effects through 5-HT3 receptor blockade in the gut [36–39]
- Small, frequent meals; consider temporary evening dosing
- Switching (preferred approach when feasible)
- If nausea persists beyond 3–4 weeks despite dose optimization and adjunctive measures, consider switching to a lower-risk agent [2,3]
- Bupropion: No serotonergic activity; minimal GI side effects
- Mirtazapine: Active antiemetic profile via 5-HT3 antagonism; associated with the fewest GI side effects among common antidepressants
- Trazodone: Nausea-neutral profile
- Agomelatine (if available): Melatonergic mechanism with minimal serotonergic GI burden
- If nausea persists beyond 3–4 weeks despite dose optimization and adjunctive measures, consider switching to a lower-risk agent [2,3]
- Pharmacological approaches (when switching is not feasible)
- Reserve for patients with a confirmed good antidepressant response on the current agent where no comparable alternative exists, or as a temporary bridge while tolerance develops
- 5-HT3 antagonists:
- Ondansetron 4–8 mg PRN (directly counteracts the serotonergic mechanism) [40,41]
- Available as orally disintegrating tablet (ODT) for faster onset; relatively affordable as a generic
- Note:
- Ondansetron is associated with QTc prolongation
- Avoid with psychotropic medications that also prolong QTc (e.g., citalopram, escitalopram, ziprasidone, thioridazine) and in patients with other QTc risk factors
- Ondansetron 4–8 mg PRN (directly counteracts the serotonergic mechanism) [40,41]
- Metoclopramide:
- Particularly effective antiemetic because it works through multiple mechanisms
- D2 receptor blockade at the area postrema (chemoreceptor trigger zone)
- 5-HT3 receptor blockade
- Prokinetic effects: increases lower esophageal sphincter tone, decreases pyloric sphincter tone, and increases peristalsis
- Can cause akathisia and (rarely) dystonia; however, the concern about tardive dyskinesia is not relevant for short-term use (days to a few weeks)
- Avoid treatment with metoclopramide tablets for longer than 12 weeks [42]
- Particularly effective antiemetic because it works through multiple mechanisms
- Mirtazapine augmentation:
- Low-dose mirtazapine (7.5–15 mg at bedtime) can be added as an adjunctive antiemetic strategy, leveraging its intrinsic 5-HT3 antagonism [12,43]
- Consider this option when there is another reason for using mirtazapine (e.g., insomnia, appetite stimulation, augmentation of antidepressant response)
- Proton pump inhibitors and H2 blockers:
- H2 blockers (e.g., famotidine) or proton pump inhibitors (e.g., omeprazole, lansoprazole) may reduce gastric acid-mediated nausea, particularly in patients with pre-existing GERD or functional dyspepsia [12]
- Cyproheptadine (H1 blocker with antiserotonergic properties) is occasionally used to manage GI hypermotility, though its use is often limited by its sedative effects [12]
- Omeprazole and esomeprazole inhibit CYP2C19 and can increase citalopram and escitalopram levels; sertraline concentrations were significantly higher in patients treated with esomeprazole [44]
- Consider pantoprazole or lansoprazole when a PPI is needed
Diarrhea

- Non-pharmacological measures
- Dietary modifications: Avoid caffeine, lactose, and high-fat foods; consider the BRAT diet temporarily
- Hydration: Ensure adequate oral fluid and electrolyte replacement
- Probiotics: Oral probiotics (e.g., Lactobacillus acidophilus) are popular nonprescription remedies, but their efficacy for psychotropically induced GI hypermotility has not been formally studied [12,41]
- Dose reduction: If clinically feasible, reducing the dose may alleviate diarrhea given the dose-dependent nature of serotonergic GI effects [3]
- Switching:
- If using sertraline, vilazodone, or fluvoxamine, consider switching to fluoxetine, citalopram, or escitalopram (lower diarrhea risk), or to a non-serotonergic agent [3]
- For patients with IBS-D who require an antidepressant, low-dose TCA (e.g., amitriptyline 10–25 mg) may simultaneously treat diarrhea and comorbid pain
- Severe or persistent diarrhea in the absence of other identified etiologies may warrant drug cessation [41]
- Pharmacological approaches
- Loperamide 2–4 mg PRN (slows intestinal motility) [41]
- Bismuth subsalicylate (Pepto-Bismol): Over-the-counter option for mild symptoms [41]
- Dietary fiber (psyllium husk–containing products): Increases stool bulk and may normalize transit [41]
- Cyproheptadine: Occasionally used to manage GI hypermotility, though limited by its sedative effects [12,41]
Constipation

- Non-pharmacological measures[45]
- Hydration: Increase daily water intake
- Dietary fiber: Emphasize fresh fruits, vegetables, bran and whole grains; fiber supplements (psyllium, methylcellulose) may be added if dietary intake is insufficient
- Physical activity: Regular exercise promotes intestinal motility
- Bowel routine: Encourage consistent timing and unhurried bathroom habits
- These measures should be recommended proactively at treatment initiation for patients prescribed TCAs or other high-constipation agents
- Switching
- If constipation persists on a TCA, consider switching to an SSRI (generally sparing for constipation, except paroxetine) [3]
- Constipation is dose-dependent for vortioxetine, consider dose reduction [2]
- Fluoxetine and sertraline are among the best-tolerated options regarding constipation risk [2]
- Pharmacological approaches
- Osmotic laxatives:
- Polyethylene glycol (PEG/MiraLAX) 17 g daily (titrate to effect; onset may take up to ~72 hours) [46,47]
- Lactulose 15–30 mL/day (cramping/distention common)
- Bulk-forming agents: Psyllium (Metamucil), methylcellulose (Citrucel) [41,45]
- Stool softeners: Docusate sodium 100–200 mg daily (may have weaker efficacy than osmotic agents [48])
- Stimulant laxatives (short-term/rescue): Bisacodyl or senna [41]
- Prokinetic agents (for refractory anticholinergic-associated constipation):
- Metoclopramide or bethanechol offer mechanistically specific strategies to counteract anticholinergic-associated constipation by stimulating peristalsis [41]
- Osmotic laxatives:
Dry mouth (xerostomia)

- Non-pharmacological measures
- Hydration and saliva stimulation: [45,49]
- Frequent sips of water
- Sugar-free gum or lozenges to stimulate salivary flow
- Avoid caffeine, alcohol, and tobacco (which worsen xerostomia)
- Saliva substitutes and protective measures: [49]
- Artificial saliva sprays or gels
- Fluoride rinses for dental protection, as dry mouth increases risk for dental caries, candidiasis, and periodontal disease [50]
- Hydration and saliva stimulation: [45,49]
- Pharmacological approaches: severe cases when switching is not an option
- Pilocarpine 10–30 mg BID or TID (cholinergic agonist; stimulates salivary secretion) [51]
- Cevimeline 30 mg three times daily (selective M3 muscarinic agonist; used in Sjögren’s syndrome)
- Dry mouth tends to persist during long-term treatment and may require active, ongoing management [12,26]
- Switching
- Vortioxetine and agomelatine are the preferred alternatives when xerostomia is the primary concern — both showed no significant dry mouth in either meta-analysis [3,52]
- Fluoxetine and citalopram are reasonable SSRI alternatives with lower dry mouth burden [3]
- Note: Mirtazapine showed significant dry mouth in the Wen analysis (OR 3.15) despite being non-significant in Oliva; caution is warranted when switching specifically for xerostomia [3,52]
When to Switch vs. When to Manage
The decision to switch antidepressants versus managing GI adverse effects conservatively depends on severity, time course, impact on adherence, and treatment response, and should be made through shared decision-making [12,53]
- Favor switching when:
- GI side effects are severe, persistent (>3–4 weeks despite optimization), or significantly impairing adherence [53]
- Nausea persists in ~32% of patients at 3 months, and diarrhea in ~45%, suggesting that early persistence is a meaningful signal [54]
- The patient is experiencing multiple GI symptoms simultaneously (e.g., nausea + diarrhea) [52]
- The current agent has not yet demonstrated clear antidepressant efficacy and tolerability is a problem [55]
- The patient identifies GI side effects as intolerable or a barrier to continuing treatment [26,55,56]
- Lower-risk alternatives with comparable efficacy are available [3,52]
- GI side effects are severe, persistent (>3–4 weeks despite optimization), or significantly impairing adherence [53]
- Favor management when:
- GI symptoms are mild-to-moderate and the patient is responding well to the antidepressant [57]
- Symptoms emerged recently (<2–3 weeks) and are expected to attenuate with tolerance [12]
- Adjunctive measures (dose adjustment, timing, pharmacological adjuncts) have not yet been exhausted [12,57]
- The patient has a history of treatment-resistant depression with limited effective options
References
1. Wang, Y., Zhang, Q., Dai, X., Xiao, G., & Luo, H. (2022). Effect of low-dose esketamine on pain control and postpartum depression after cesarean section: A retrospective cohort study. Annals of Palliative Medicine, 11(1), 45–57. https://doi.org/10.21037/apm-21-3343
2. Oliva, V., Lippi, M., Paci, R., Del Fabro, L., Delvecchio, G., Brambilla, P., De Ronchi, D., Fanelli, G., & Serretti, A. (2021). Gastrointestinal side effects associated with antidepressant treatments in patients with major depressive disorder: A systematic review and meta-analysis. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 109, 110266. https://doi.org/10.1016/j.pnpbp.2021.110266
3. Wen, S., Yan, Y., Shao, J., Xie, H., Wang, M., Dou, Y., Liu, D., Yang, X., & Ma, X. (2025). Comparative gastrointestinal effects of antidepressants for the acute treatment of adults with major depressive disorder: A network and dose‒response meta-analysis. Translational Psychiatry, 16, 34. https://doi.org/10.1038/s41398-025-03751-3
4. Gartlehner, G., Hansen, R. A., Reichenpfader, U., Kaminski, A., Kien, C., Strobelberger, M., Noord, M. V., Thieda, P., Thaler, K., & Gaynes, B. (2011). Drug Class Review: Second-Generation Antidepressants. Oregon Health & Science University. https://www.ncbi.nlm.nih.gov/books/NBK54355/
5. Gosmann, N. P., Costa, M. de A., Jaeger, M. de B., Frozi, J., Spanemberg, L., Manfro, G. G., Cortese, S., Cuijpers, P., Pine, D. S., & Salum, G. A. (2023). Incidence of adverse events and comparative tolerability of selective serotonin reuptake inhibitors, and serotonin and norepinephrine reuptake inhibitors for the treatment of anxiety, obsessive-compulsive, and stress disorders: A systematic review and network meta-analysis. Psychological Medicine, 53(9), 3783–3792. https://doi.org/10.1017/S0033291723001630
6. Cepeda, M. S., Katz, E. G., & Blacketer, C. (2017). Microbiome-Gut-Brain Axis: Probiotics and Their Association With Depression. The Journal of Neuropsychiatry and Clinical Neurosciences, 29(1), 39–44. https://doi.org/10.1176/appi.neuropsych.15120410
7. Collins, S. M. (2020). Interrogating the Gut-Brain Axis in the Context of Inflammatory Bowel Disease: A Translational Approach. Inflammatory Bowel Diseases, 26(4), 493–501. https://doi.org/10.1093/ibd/izaa004
8. Gershon, M. D. (2013). 5-Hydroxytryptamine (serotonin) in the gastrointestinal tract. Current Opinion in Endocrinology, Diabetes, and Obesity, 20(1), 14–21. https://doi.org/10.1097/MED.0b013e32835bc703
9. Israelyan, N., Colle, A. D., Li, Z., Park, Y., Xing, A., Jacobsen, J. P. R., Luna, R. A., Jensen, D. D., Madra, M., Saurman, V., Rahim, R., Latorre, R., Law, K., Carson, W., Bunnett, N. W., Caron, M. G., & Margolis, K. G. (2019). Effects of Serotonin and Slow-release 5-HTP on Gastrointestinal Motility in a Mouse Model of Depression. Gastroenterology, 157(2), 507–521.e4. https://doi.org/10.1053/j.gastro.2019.04.022
10. Wang, Z., Li, H., Kang, Y., Liu, Y., Shan, L., & Wang, F. (2022). Risks of Digestive System Side-Effects of Selective Serotonin Reuptake Inhibitors in Patients with Depression: A Network Meta-Analysis. Therapeutics and Clinical Risk Management, 18, 799–812. https://doi.org/10.2147/TCRM.S363404
11. Zhong, W., Shahbaz, O., Teskey, G., Beever, A., Kachour, N., Venketaraman, V., & Darmani, N. A. (2021). Mechanisms of Nausea and Vomiting: Current Knowledge and Recent Advances in Intracellular Emetic Signaling Systems. International Journal of Molecular Sciences, 22(11), 5797. https://doi.org/10.3390/ijms22115797
12. Kelly, K., Posternak, M., & Jonathan, E. A. (2008). Toward achieving optimal response: Understanding and managing antidepressant side effects. Dialogues in Clinical Neuroscience, 10(4), 409–418. https://doi.org/10.31887/DCNS.2008.10.4/kkelly
13. Ostenfeld, A., Petersen, T. S., Pedersen, L. H., Westergaard, H. B., Løkkegaard, E. C. L., & Andersen, J. T. (2022). Mirtazapine exposure in pregnancy and fetal safety: A nationwide cohort study. Acta Psychiatrica Scandinavica, 145(6), 557–567. https://doi.org/10.1111/acps.13431
14. Coskun, M., & Alyanak, B. (2011). Psychiatric Co-morbidity and Efficacy of Mirtazapine Treatment in Young Subjects With Chronic or Cyclic Vomiting Syndromes: A Case Series. Journal of Neurogastroenterology and Motility, 17(3), 305–311. https://doi.org/10.5056/jnm.2011.17.3.305
15. Bétry, C., Etiévant, A., Oosterhof, C., Ebert, B., Sanchez, C., & Haddjeri, N. (2011). Role of 5-HT3 Receptors in the Antidepressant Response. Pharmaceuticals, 4(4), 603–629. https://doi.org/10.3390/ph4040603
16. Hoffman, J. M., Tyler, K., MacEachern, S. J., Balemba, O. B., Johnson, A. C., Brooks, E. M., Zhao, H., Swain, G. M., Moses, P. L., Galligan, J. J., Sharkey, K. A., Greenwood-Van Meerveld, B., & Mawe, G. M. (2012). Activation of colonic mucosal 5-HT(4) receptors accelerates propulsive motility and inhibits visceral hypersensitivity. Gastroenterology, 142(4), 844–854.e4. https://doi.org/10.1053/j.gastro.2011.12.041
17. Mawe, G. M., & Hoffman, J. M. (2013). Serotonin Signaling in the Gastrointestinal Tract: Nature Reviews. Gastroenterology & Hepatology, 10(8), 473–486. https://doi.org/10.1038/nrgastro.2013.105
18. Acharekar, M. V., Guerrero Saldivia, S. E., Unnikrishnan, S., Chavarria, Y. Y., Akindele, A. O., Jalkh, A. P., Eastmond, A. K., Shetty, C., Rizvi, S. M. H. A., Sharaf, J., Williams, K.-A. D., Tariq, M., & Balani, P. (n.d.). A Systematic Review on the Efficacy and Safety of Selective Serotonin Reuptake Inhibitors in Gastrointestinal Motility Disorders: More Control, Less Risk. Cureus, 14(8), e27691. https://doi.org/10.7759/cureus.27691
19. Cates, A. N. de, Harmer, C. J., Harrison, P. J., Cowen, P. J., Emmanuel, A., Travis, S., Murphy, S. E., & Taquet, M. (2024). Association between a selective 5-HT4 receptor agonist and incidence of major depressive disorder: Emulated target trial. The British Journal of Psychiatry, 225(3), 371–378. https://doi.org/10.1192/bjp.2024.97
20. Brambilla, P., Cipriani, A., Hotopf, M., & Barbui, C. (2005). Side-effect profile of fluoxetine in comparison with other SSRIs, tricyclic and newer antidepressants: A meta-analysis of clinical trial data. Pharmacopsychiatry, 38(2), 69–77. https://doi.org/10.1055/s-2005-837806
21. Remick, R. A. (1988). Anticholinergic side effects of tricyclic antidepressants and their management. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 12(2–3), 225–231. https://doi.org/10.1016/0278-5846(88)90039-5
22. Ghossein, N., Kang, M., & Lakhkar, A. D. (2025). Anticholinergic Medications. In StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/books/NBK555893/
23. Moore, P. A., & Guggenheimer, J. (2008). Medication-induced hyposalivation: Etiology, diagnosis, and treatment. Compendium of Continuing Education in Dentistry, 29(1), 50–55. https://www.ncbi.nlm.nih.gov/pubmed/18361341
24. Cangemi, D. J., & Kuo, B. (2019). Practical Perspectives in the Treatment of Nausea and Vomiting. Journal of Clinical Gastroenterology, 53(3), 170–178. https://doi.org/10.1097/MCG.0000000000001164
25. Gill, H., Gill, B., El-Halabi, S., Chen-Li, D., Lipsitz, O., Rosenblat, J. D., Van Rheenen, T. E., Rodrigues, N. B., Mansur, R. B., Majeed, A., Lui, L. M. W., Nasri, F., Lee, Y., & Mcintyre, R. S. (2020). Antidepressant Medications and Weight Change: A Narrative Review. Obesity, 28(11), 2064–2072. https://doi.org/10.1002/oby.22969
26. Niarchou, E., Roberts, L., & Naughton, B. D. (2024). What is the impact of antidepressant side effects on medication adherence among adult patients diagnosed with depressive disorder: A systematic review. Journal of Psychopharmacology (Oxford, England), 38(2), 127–136. https://doi.org/10.1177/02698811231224171
27. Moraczewski, J., Awosika, A. O., & Aedma, K. K. (2023). Tricyclic Antidepressants. In StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/books/NBK557791/
28. Arany, S., Kopycka-Kedzierawski, D. T., Caprio, T. V., & Watson, G. E. (2021). Anticholinergic medication: Related dry mouth and effects on the salivary glands. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology, 132(6), 662–670. https://doi.org/10.1016/j.oooo.2021.08.015
29. Talley, N. J., Locke, G. R., Saito, Y. A., Almazar, A. E., Bouras, E. P., Howden, C. W., Lacy, B. E., DiBaise, J. K., Prather, C. M., Abraham, B. P., El-Serag, H. B., Moayyedi, P., Herrick, L. M., Szarka, L. A., Camilleri, M., Hamilton, F. A., Schleck, C. D., Tilkes, K. E., & Zinsmeister, A. R. (2015). Effect of Amitriptyline and Escitalopram on Functional Dyspepsia: A Multi-Center, Randomized, Controlled Study. Gastroenterology, 149(2), 340–349.e2. https://doi.org/10.1053/j.gastro.2015.04.020
30. Shanmugham, S., Zuber, M., Chan, J. E., Kumar, S., Ching, S. M., Lee, Y. Y., Vadakkechalil, H., & Veettil, S. K. (2025). Efficacy of antidepressants in functional dyspepsia: Systematic review and meta-analysis with trial sequential analysis of randomized controlled trials. Indian Journal of Gastroenterology: Official Journal of the Indian Society of Gastroenterology, 44(1), 24–34. https://doi.org/10.1007/s12664-024-01648-5
31. Torta, R. G. (2012). Antidepressant up-titration: Pharmacological and psychological considerations. Expert Opinion on Drug Safety, 11(5), 685–688. https://doi.org/10.1517/14740338.2012.712683
32. Carvalho, A. F., Sharma, M. S., Brunoni, A. R., Vieta, E., & Fava, G. A. (2016). The Safety, Tolerability and Risks Associated with the Use of Newer Generation Antidepressant Drugs: A Critical Review of the Literature. Psychotherapy and Psychosomatics, 85(5), 270–288. https://doi.org/10.1159/000447034
33. DeVane, C. L. (2003). Immediate-release versus controlled-release formulations: Pharmacokinetics of newer antidepressants in relation to nausea. The Journal of Clinical Psychiatry, 64 Suppl 18, 14–19. https://www.ncbi.nlm.nih.gov/pubmed/14700450
34. Nussbaumer, B., Morgan, L. C., Reichenpfader, U., Greenblatt, A., Hansen, R. A., Van Noord, M., Lux, L., Gaynes, B. N., & Gartlehner, G. (2014). Comparative efficacy and risk of harms of immediate- versus extended-release second-generation antidepressants: A systematic review with network meta-analysis. CNS Drugs, 28(8), 699–712. https://doi.org/10.1007/s40263-014-0169-z
35. Meade. (2022). Predicting Drug Absorption in Patients with a Short Bowel.
36. Ataei, S., Jahangard, L., Anaraki, S., Dastan, D., & Khazaei, S. (2025). Investigating the Effect of Ginger as an Adjunctive Treatment along with SSRIs to Reduce Anxiety in Patients with GAD: A Clinical Trial Study. Current Psychiatry Research and Reviews, 21(2), 177–185. https://doi.org/10.2174/0126660822258340231025171755
37. Current Psychiatry. 2003 February;02(02):78-78. (2003). Using ginger to treat antidepressant-induced nausea | MDedge. https://mdedge.com/psychiatry/article/59696/depression/using-ginger-treat-antidepressant-induced-nausea
38. Marx, W., Ried, K., McCarthy, A. L., Vitetta, L., Sali, A., McKavanagh, D., & Isenring, L. (2017). Ginger-Mechanism of action in chemotherapy-induced nausea and vomiting: A review. Critical Reviews in Food Science and Nutrition, 57(1), 141–146. https://doi.org/10.1080/10408398.2013.865590
39. Lete, I., & Allué, J. (2016). The Effectiveness of Ginger in the Prevention of Nausea and Vomiting during Pregnancy and Chemotherapy. Integrative Medicine Insights, 11, 11–17. https://doi.org/10.4137/IMI.S36273
40. Calabrese, L., & McEnany, G. (2001). Ondansetron in the Treatment of Antidepressant-Induced Nausea. Journal of the American Psychiatric Nurses Association, 7(3), 94–96. https://doi.org/10.1067/mpn.2001.115883
41. Goldberg, J. F., & Ernst, C. L. (2018). Managing the Side Effects of Psychotropic Medications (2nd ed.). American Psychiatric Association Publishing.
42. Isola, S., Hussain, A., Dua, A., Singh, K., & Adams, N. (2025). Metoclopramide. In StatPearls. StatPearls Publishing. http://www.ncbi.nlm.nih.gov/books/NBK519517/
43. Pae, C.-U. (2006). Low-dose mirtazapine may be successful treatment option for severe nausea and vomiting. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 30(6), 1143–1145. https://doi.org/10.1016/j.pnpbp.2006.03.015
44. Gjestad, C., Westin, A. A., Skogvoll, E., & Spigset, O. (2015). Effect of Proton Pump Inhibitors on the Serum Concentrations of the Selective Serotonin Reuptake Inhibitors Citalopram, Escitalopram, and Sertraline. Therapeutic Drug Monitoring, 37(1), 90–97. https://doi.org/10.1097/FTD.0000000000000101
45. Staff, M. C. (2019). Antidepressants: Get tips to cope with side effects. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/depression/in-depth/antidepressants/art-20049305
46. Sadler, K., Arnold, F., & Dean, S. (2022). Chronic Constipation in Adults. American Family Physician, 106(3), 299–306. https://www.aafp.org/pubs/afp/issues/2022/0900/chronic-constipation-adults.html
47. Chang, L., Chey, W. D., Imdad, A., Almario, C. V., Bharucha, A. E., Diem, S., Greer, K. B., Hanson, B., Harris, L. A., Ko, C., Murad, M. H., Patel, A., Shah, E. D., Lembo, A. J., & Sultan, S. (2023). American Gastroenterological Association-American College of Gastroenterology Clinical Practice Guideline: Pharmacological Management of Chronic Idiopathic Constipation. Gastroenterology, 164(7), 1086–1106. https://doi.org/10.1053/j.gastro.2023.03.214
48. Tarumi, Y., Wilson, M. P., Szafran, O., & Spooner, G. R. (2013). Randomized, double-blind, placebo-controlled trial of oral docusate in the management of constipation in hospice patients. Journal of Pain and Symptom Management, 45(1), 2–13. https://doi.org/10.1016/j.jpainsymman.2012.02.008
49. Daly, C. (2016). Oral and dental effects of antidepressants. Australian Prescriber, 39(3), 84. https://doi.org/10.18773/austprescr.2016.035
50. Wright, C. (2012). Therapeutic Guidelines: Oral and Dental. Version 2. Australian Prescriber, 35(6). https://doi.org/10.18773/austprescr.2012.089
51. Masters, K. J. (2005). Pilocarpine treatment of xerostomia induced by psychoactive medications. The American Journal of Psychiatry, 162(5), 1023. https://doi.org/10.1176/appi.ajp.162.5.1023
52. Oliva, V., Lippi, M., Paci, R., Del Fabro, L., Delvecchio, G., Brambilla, P., De Ronchi, D., Fanelli, G., & Serretti, A. (2021). Gastrointestinal side effects associated with antidepressant treatments in patients with major depressive disorder: A systematic review and meta-analysis. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 109, 110266. https://doi.org/10.1016/j.pnpbp.2021.110266
53. Gautam, S., Jain, A., Gautam, M., Vahia, V., & Grover, S. (2017). Clinical Practice Guidelines for the management of Depression. Indian Journal of Psychiatry, 59(5), 34. https://doi.org/10.4103/0019-5545.196973
54. Kelly, K., Posternak, M., & Jonathan, E. A. (2008). Toward achieving optimal response: Understanding and managing antidepressant side effects. Dialogues in Clinical Neuroscience, 10(4), 409–418. https://doi.org/10.31887/DCNS.2008.10.4/kkelly
55. Kennedy, S. H., Lam, R. W., McIntyre, R. S., Tourjman, S. V., Bhat, V., Blier, P., Hasnain, M., Jollant, F., Levitt, A. J., MacQueen, G. M., McInerney, S. J., McIntosh, D., Milev, R. V., Müller, D. J., Parikh, S. V., Pearson, N. L., Ravindran, A. V., & Uher, R. (2016). Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive Disorder. Canadian Journal of Psychiatry. Revue Canadienne de Psychiatrie, 61(9), 540–560. https://doi.org/10.1177/0706743716659417
56. Trindade, E., Menon, D., Topfer, L. A., & Coloma, C. (1998). Adverse effects associated with selective serotonin reuptake inhibitors and tricyclic antidepressants: A meta-analysis. CMAJ: Canadian Medical Association Journal, 159(10), 1245–1252. https://pmc.ncbi.nlm.nih.gov/articles/PMC1229819/
57. Goldberg, J. F., & Ernst, C. L. (2019). Managing the side effects of psychotropic medications (Second edition). American Psychiatric Association Publishing.



