Q1 2026 in Review
In a nutshell
Q1 2026 brought two FDA actions and one clinical guideline update. The FDA approved milsaperidone and removed the suicidality warning from GLP-1 receptor agonists. The ACOG issued updated guidance positioning zuranolone as the oral option for severe postpartum depression, now that brexanolone has been withdrawn from the market.
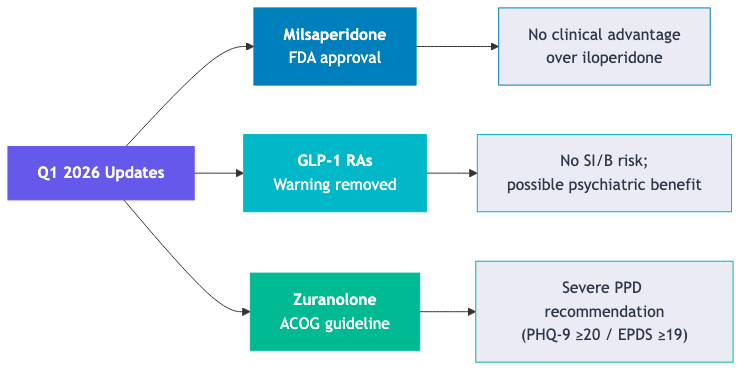
- Milsaperidone: a “new” antipsychotic without a compelling clinical case
- Milsaperidone is an active metabolite of iloperidone; the two interconvert in vivo and share the same efficacy, safety, and tolerability profile
- There is no evidence-based clinical reason to prescribe milsaperidone over iloperidone as of March 2026
- GLP-1 RAs: reassure patients
- The suicidality warning originated from older weight-loss drugs, not from GLP-1 RA-specific data; it has now been removed
- The FDA’s review found no increased risk of suicidal ideation, anxiety, depression, or psychosis
- A 2026 national cohort study suggests semaglutide may reduce the worsening of depression, anxiety, and substance use disorder. RCTs are needed
- Zuranolone: ACOG’s updated guidance after brexanolone’s withdrawal
- With brexanolone no longer available, ACOG issued a 2026 Clinical Practice Update positioning zuranolone as the oral alternative for severe PPD (PHQ-9 ≥20 or EPDS ≥19)
- Practical requirements: 14-day course, once daily at bedtime with a fatty meal; patients must not care for their infant alone for 12 hours after each dose
- Limitations:
- No head-to-head data vs. SSRIs, SNRIs, or psychotherapy; efficacy data extend only to day 45
Milsaperidone (Bysanti) Approved for Schizophrenia and Bipolar Disorder
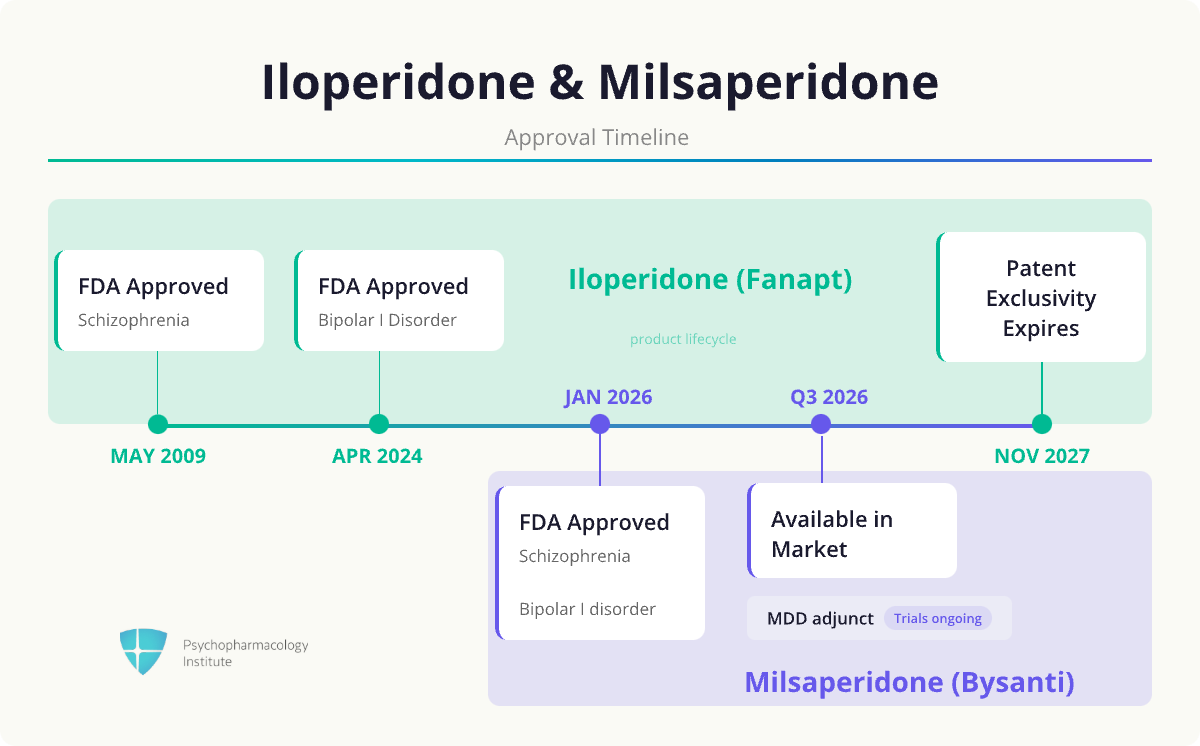
- The FDA approved Milsaperidone (Bysanti) in January 2026 for the treatment of schizophrenia in adults and bipolar I disorder mania and mixed mania [1]
- What is milsaperidone?
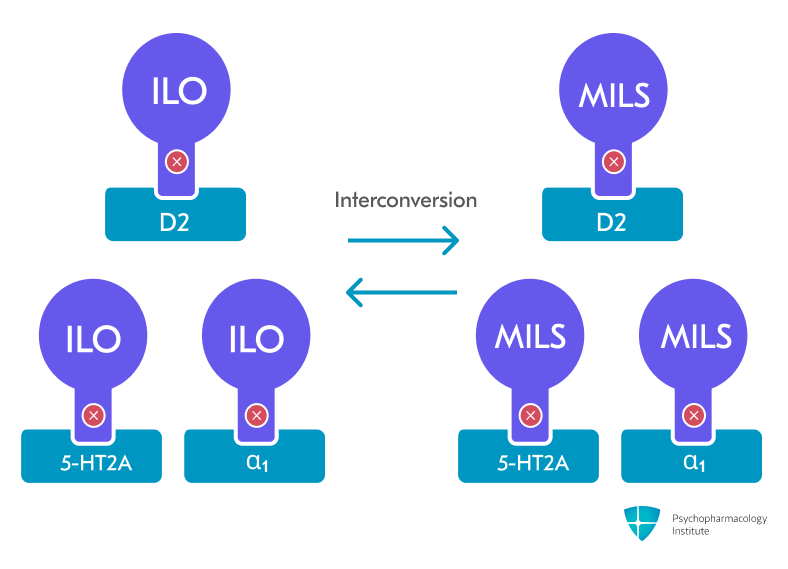
- Milsaperidone is an active metabolite of iloperidone
- It has the same pharmacodynamic properties as iloperidone
- Pharmacology
- Milsaperidone and iloperidone rapidly interconvert in vivo
- When milsaperidone is administered orally, it converts to iloperidone
- Pharmacodynamics [2,3]
- 5-HT2A antagonist
- Alpha-1 antagonist
- D2 antagonist
- Milsaperidone and iloperidone rapidly interconvert in vivo
- Regulatory aspects: New chemical entity (NCE) [1,4]
- Milsaperidone was granted NCE status: it borrows iloperidone’s entire clinical data package to establish efficacy while carrying independent patent protection through 2044
- Bioequivalent to iloperidone across the full therapeutic dose range
- Approved via the 505(b)(2) pathway: allows a new application to rely on existing clinical data from a previously approved drug, without running new efficacy trials
- The efficacy basis came entirely from iloperidone’s clinical program
- Vanda submitted pharmacokinetic bioequivalence data, not new Phase III trials
- Practical aspects
- Titration required:
- Iloperidone and milsaperidone require gradual titration over several days to mitigate orthostatic hypotension and syncope risk
- Twice-daily dosing
- Twice-daily dosing is a known adherence barrier in chronic psychiatric conditions [5]
- Titration required:
- Side effects
- Both drugs carry a risk of QTc prolongation.
- This places them among the higher-risk atypical antipsychotics for QTc, comparable to ziprasidone [6]
- Both drugs carry a risk of QTc prolongation.
- Commercial context
- Iloperidone patent exclusivity expires in November 2027; three manufacturers have already filed.
- Milsaperidone’s NCE designation extends Vanda’s commercial position ahead of iloperidone’s patent expiration in November 2027
- Patent protection runs through 2044, providing a protected asset for pipeline development
- In the pipeline [1]
- LAI formulation
- Milsaperidone’s physicochemical properties are amenable to lipid ester development for long-acting injectable (LAI) formulations
- Currently in preclinical/early development
- MDD as once-daily dosing
- Milsaperidone is currently being tested as a once-daily adjunctive treatment in treatment-resistant major depressive disorder
- The ongoing clinical study is expected to be completed by the end of 2026
- LAI formulation
- Should we prescribe milsaperidone over iloperidone?
- There is currently no evidence-based clinical reason for prescribing milsaperidone over iloperidone
- Milsaperidone carries the same efficacy, safety profile, and formulation aspects as iloperidone as of March 2026.
- Why is Vanda pursuing milsaperidone if it offers no current clinical advantage over iloperidone?
- Milsaperidone’s approval can be better characterized as a regulatory platform
- By establishing milsaperidone as a new chemical entity before iloperidone loses exclusivity, Vanda secures an IP runway for an LAI formulation and a once-daily MDD indication
- Both indications are technically feasible under iloperidone, but commercially unviable with only ~18 months of exclusivity remaining before generic entry
GLP-1 RA Suicidality Warning Removed
- In January 2026, the FDA requested that manufacturers remove the suicidal ideation and behavior (SI/B) warning from the labeling of GLP-1 receptor agonists [7]
- Affected products: Saxenda (liraglutide), Wegovy (semaglutide), and Zepbound (tirzepatide) [8]
- Implications:
- GLP-1 agonists no longer carry a suicidal ideation warning
- Patients can be reassured that the FDA found no increased risk of suicidal ideation and behavior after a comprehensive review of the available data
- Background
- The suicidal ideation and behavior (SI/B) warning had been included in the GLP-1 RA labeling at the time of original FDA approvals
- The warning was not based on GLP-1 RA-specific data
- It was carried over from postmarketing reports of SI/B observed with a variety of older medicines used or studied for weight loss
- A confounding factor: patients with obesity and diabetes carry elevated baseline rates of depression and anxiety [9]
- Earlier observational studies raising psychiatric concerns about GLP-1 RAs were likely affected by this underlying burden
- GLP-1 agonists approved for glycemic control in type 2 diabetes had never included this warning
- Regulatory history
- July 2023: FDA initiated an investigation after receiving postmarketing reports of SI/B in GLP-1 RA users
- January 2024: FDA issued a preliminary Drug Safety Communication; initial review did not find an association, but uncertainty remained due to small SI/B case numbers in individual trials
- January 2026: FDA completes comprehensive review and requests label removal
- What the FDA found
- No increased risk of: suicidal ideation and behavior, anxiety, depression, irritability, or psychosis
- Methods:
- Meta-analysis of 91 placebo-controlled trials, including 107,910 patients (60,338 on a GLP-1 RA; 47,572 on placebo)
- Retrospective cohort study using healthcare claims data comparing intentional self-harm risk between GLP-1 RA users and SGLT2 inhibitor users
- Review of published observational and pooled studies
- Emerging evidence: potential psychiatric benefits
- A 2026 national cohort study goes further than ruling out harm [9]
- Key findings:
- Semaglutide was associated with a 42% lower risk of worsening mental illness and liraglutide with an 18% lower risk, compared with non-use periods in the same individuals
- Semaglutide was specifically associated with reduced worsening of depression , anxiety, and substance use disorder
- Causality cannot be established from observational data; randomized controlled trials are needed to confirm these findings
- Key findings:
- A 2026 national cohort study goes further than ruling out harm [9]
- Implications of the full evidence base
- The available data do not support earlier concerns about GLP-1 RAs worsening psychiatric outcomes
- Semaglutide may have meaningful benefits for mood and anxiety in patients with comorbid diabetes or obesity
- This raises the possibility of genuine psychiatric benefit, pending confirmation from randomized controlled trials
Zuranolone for Severe PPD: ACOG’s 2026 Clinical Practice Update
- In January 2026, the American College of Obstetricians and Gynecologists (ACOG) issued a Clinical Practice Update on zuranolone (Zurzuvae)
- Recommendation: [10]
- Consider zuranolone for severe postpartum depression
- Onset: third trimester or within 4 weeks postpartum
- Eligibility window: within 12 months of delivery
- Consider zuranolone for severe postpartum depression
- On the severity recommendation
- The zuranolone prescribing information carries no severity qualifier
- ACOG’s restriction to severe PPD reflects trial design: pivotal trials required HAMD-17 >26 (consistent with severe disease)
- Efficacy in milder presentations is unestablished
- Clinical threshold: PHQ-9 ≥20 or EPDS ≥19
- Pharmacology
- Zuranolone is a synthetic neuroactive steroid and positive allosteric modulator (PAM) of GABA-A receptors [11]
- It shares its mechanism of action with brexanolone (Zulresso), which was the IV predecessor
- Zuranolone evidence limitations [12,13]
- No head-to-head data against SSRIs, SNRIs, or psychotherapy
- Comparative effectiveness vs. the standard of care is unknown
- Efficacy data extend only to day 45 (4 weeks post-treatment); no data beyond that window
- No head-to-head data against SSRIs, SNRIs, or psychotherapy
- FDA Approval, Scheduling, and the Brexanolone Exit
- Zuranolone (Zurzuvae) received FDA approval in August 2023 for postpartum depression in adults
- Schedule IV controlled substance
- The FDA identified benzodiazepine-like reinforcing effects, dose-dependent euphoric effects, and biochemical similarity to barbiturates
- Brexanolone (Zulresso): first FDA-approved treatment specifically for PPD (2019), sharing zuranolone’s GABA-A PAM mechanism [14]
- Required a 60-hour continuous inpatient IV infusion under a REMS program
- Complex logistics and high cost severely limited real-world access
- Sage Therapeutics withdrew FDA approval on April 14, 2025, pivoting toward zuranolone as the oral successor
- Practical aspects [15]
- 14-day treatment course, not continuous chronic therapy
- Once-daily dosing at bedtime with a fatty meal (400–1,000 kcal, 25–50% fat)
- Can be used as monotherapy or adjunct to stable SSRI/SNRI therapy
- Side effects
- CNS depression: somnolence, dizziness, confusion. Most clinically significant adverse effects
- Patients should not care for their infant alone (feeding, changing, bathing) for 12 hours post-dose
- Embryo-fetal toxicity: effective contraception required during the 14-day course and for 1 week after the final dose
References
1. Vanda Pharmaceuticals Inc. (2026). Vanda Pharmaceuticals Announces FDA Approval of BYSANTI™ (milsaperidone) for the Treatment of Bipolar I Disorder and Schizophrenia — A New Chemical Entity Opening New Horizons in Psychiatric Innovation. https://www.prnewswire.com/news-releases/vanda-pharmaceuticals-announces-fda-approval-of-bysanti-milsaperidone-for-the-treatment-of-bipolar-i-disorder-and-schizophrenia—a-new-chemical-entity-opening-new-horizons-in-psychiatric-innovation-302693941.html
2. Vanda Pharmaceuticals Inc. (2026). BYSANTI (milsaperidone) Tablets Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2026/220358Orig1s000lbl.pdf
3. Vanda Pharmaceuticals Inc. (2026). FANAPT (iloperidone) Tablets Prescribing Information. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=022192
4. U.S. Food and Drug Administration. (n.d.). 21 CFR § 314.108 — New Drug Product Exclusivity. Retrieved March 22, 2026, from https://www.law.cornell.edu/cfr/text/21/314.108
5. Medic, G., Higashi, K., Littlewood, K. J., Diez, T., Granström, O., & Kahn, R. S. (2013). Dosing Frequency and Adherence in Chronic Psychiatric Disease: Systematic Review and Meta-Analysis. Neuropsychiatric Disease and Treatment, 9, 119–131. https://doi.org/10.2147/NDT.S39303
6. Woosley, R. L., Romero, K. A., Gallo, T., Woosley, J., & Darpo, B. (2013). A Thorough QTc Study of 3 Doses of Iloperidone Including Metabolic Inhibition via CYP2D6 and/or CYP3A4 and a Comparison to Quetiapine and Ziprasidone. Journal of Clinical Pharmacology, 53(3), 289–295. https://doi.org/10.1177/0091270012442192
7. U.S. Food and Drug Administration. (2026). FDA Requests Removal of Suicidal Behavior and Ideation Warning from Glucagon-Like Peptide-1 Receptor Agonist (GLP-1 RA) Medications. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-removal-suicidal-behavior-and-ideation-warning-glucagon-peptide-1-receptor-agonist-glp
8. Novo Nordisk. (2026). SAXENDA (liraglutide) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/206321s022lbl.pdf
9. Taipale, H., Taylor, M., Lähteenvuo, M., Mittendorfer-Rutz, E., Tanskanen, A., & Tiihonen, J. (2026). Association between GLP-1 receptor agonist use and worsening mental illness in people with depression and anxiety in sweden: A national cohort study. The Lancet Psychiatry, 13, 119–131. https://doi.org/10.1016/S2215-0366(26)00014-3
10. Moore Simas, T. A., Hoffman, M. C., Roussos-Ross, K., Miller, E. S., Gandhi, M., & Shields, A. (2026). Zuranolone and Brexanolone for the Treatment of Postpartum Depression. Obstetrics & Gynecology, 147(1), e24–e28.
11. Sharma, R., Bansal, P., Saini, L., Sharma, N., & Dhingra, R. (2024). Zuranolone, a Neuroactive Drug, Used in the Treatment of Postpartum Depression by Modulation of GABA(A) Receptors. Pharmacology, Biochemistry, and Behavior, 238, 173734. https://doi.org/10.1016/j.pbb.2024.173734
12. Deligiannidis, K. M., Meltzer-Brody, S., Maximos, B., Peeper, E. Q., Freeman, M., Lasser, R., Bullock, A., Kotecha, M., Li, S., Forrestal, F., Rana, N., Garcia, M., Leclair, B., & Doherty, J. (2023). Zuranolone for the Treatment of Postpartum Depression. The American Journal of Psychiatry, 180(9), 668–675. https://doi.org/10.1176/appi.ajp.20220785
13. Deligiannidis, K. M., Meltzer-Brody, S., Gunduz-Bruce, H., Doherty, J., Jonas, J., Li, S., Sankoh, A. J., Silber, C., Campbell, A. D., Werneburg, B., Kanes, S. J., & Lasser, R. (2021). Effect of Zuranolone vs Placebo in Postpartum Depression: A Randomized Clinical Trial. JAMA Psychiatry, 78(9), 951–959. https://doi.org/10.1001/jamapsychiatry.2021.1559
14. Sage Therapeutics, Inc. (2025). Withdrawal of Approval of a New Drug Application for Zulresso (Brexanolone) Solution, 100 Milligrams/20 Milliliters. https://www.federalregister.gov/documents/2025/03/14/2025-04101/sage-therapeutics-inc-withdrawal-of-approval-of-a-new-drug-application-for-zulresso-brexanolone
15. Biogen MA Inc. (2026). ZURZUVAE (zuranolone) Capsules Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/217369s001lbl.pdf



