Slides and Transcript
Slide 2 of 13
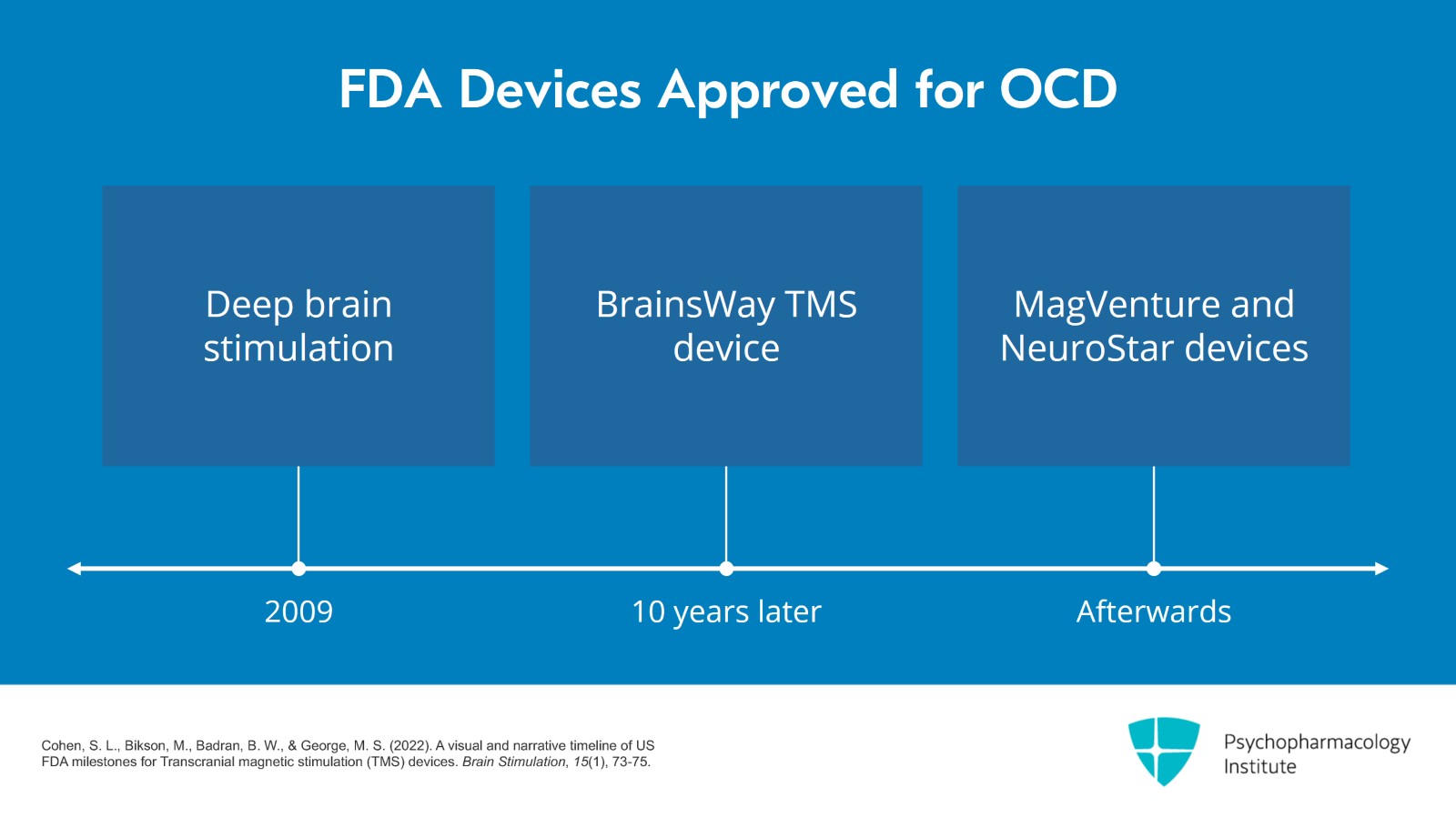
These are the devices which are FDA approved for OCD. The first was in February 2009 and that was an invasive treatment called Deep Brain Stimulation with a Humanitarian Device Exemption. Then about 10 years later, the first noninvasive treatment for OCD was approved and that was the BrainsWay TMS device. Afterwards, both MagVenture and NeuroStar got their devices approved based on equivalency to the BrainsWay device.
References:
- Cohen, S. L., Bikson, M., Badran, B. W., & George, M. S. (2022). A visual and narrative timeline of US FDA milestones for Transcranial magnetic stimulation (TMS) devices. Brain Stimulation, 15(1), 73-75.
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 3 of 13
Note that the official FDA approval actually states that the TMS devices are approved as an adjunctive treatment for OCD and you'll see why in the next slides.
References:
- Cohen, S. L., Bikson, M., Badran, B. W., & George, M. S. (2022). A visual and narrative timeline of US FDA milestones for Transcranial magnetic stimulation (TMS) devices. Brain Stimulation, 15(1), 73-75.
Slide 4 of 13
The BrainsWay device approved for OCD uses the H7 coil which targets the dorsomedial prefrontal cortex and anterior cingulate cortex more than the H1 coil which targets the cortical and subcortical prefrontal cortex structures and more on the left.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 5 of 13
Their pivotal study was 29 treatments over six weeks and used the Yale-Brown Obsessive-Compulsive Disorder Scale to assess OCD symptoms weekly until the end of week 6 and then at the end of week 10.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Slide 6 of 13
Treatments were using the Deep TMS protocol of 20 Hz stimulations for 2000 pulses at 100% of motor threshold amounting to 18 minutes each day.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 7 of 13
The very interesting aspect of the study was that before the daily treatments began the patient had to have their OCD symptoms provoked to a distress level between 4 to 7 out of 10 with 10 being worst. So that usually took 10 to 20 minutes before each session. Thus, treatment of OCD with TMS is not just about turning the machine on, it's also about the psychotherapy part which is why the official FDA indication is for TMS to be an adjunctive treatment because there's also an expectation of that psychotherapy part.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Slide 8 of 13
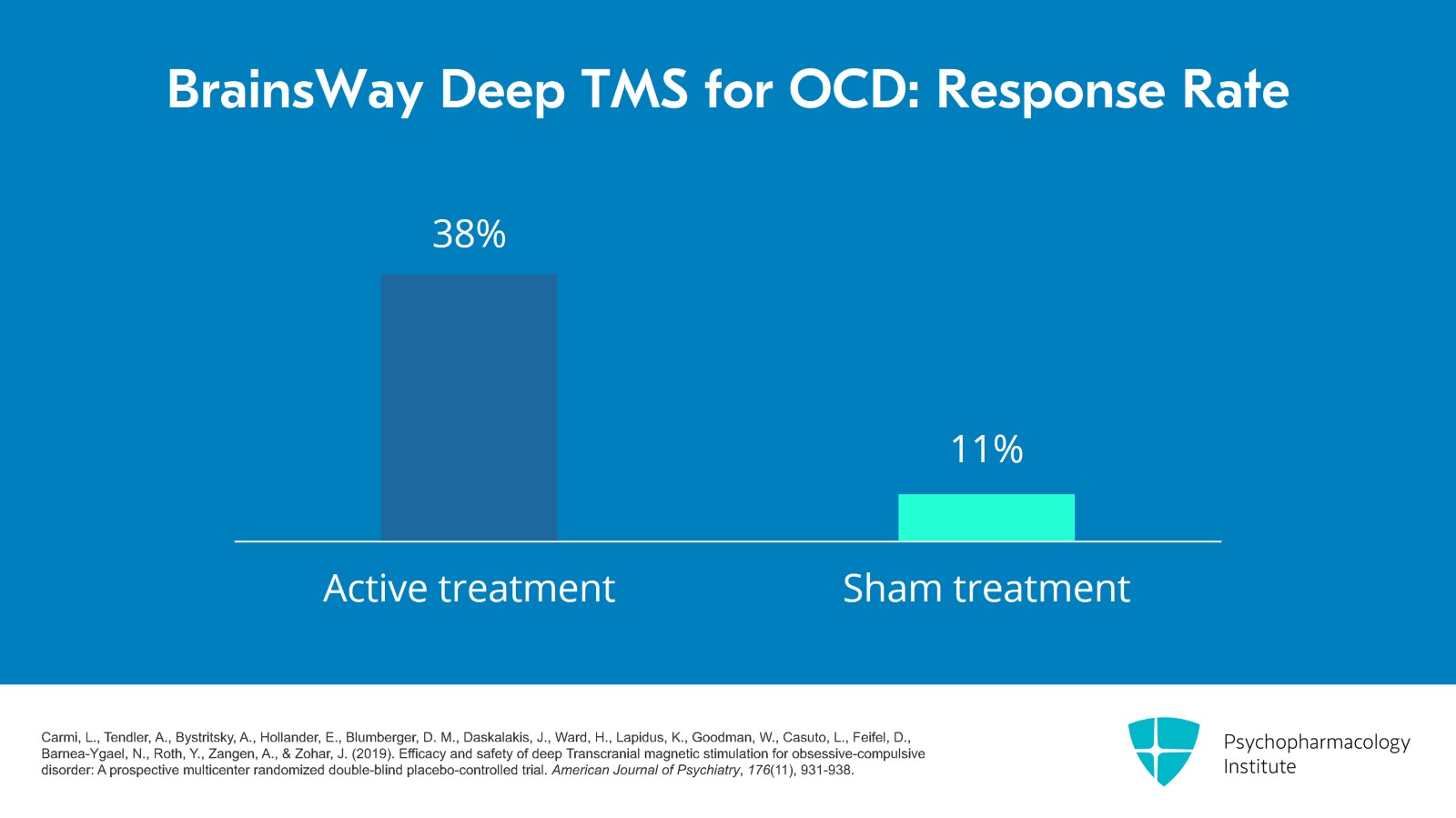
So their study enrolled 94 patients, 47 in each arm. Already at week 2, there was significant changes in the Y-BOCS measurement of OCD symptoms. After six weeks, the response rate was 38.1% for active versus 11% for sham. Note that for OCD studies, the response rate is a reduction of at least 30% of Y-BOCS, so that's a smaller amount than the depression studies where the response rate is defined as at least 50% improvement in symptoms.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 9 of 13
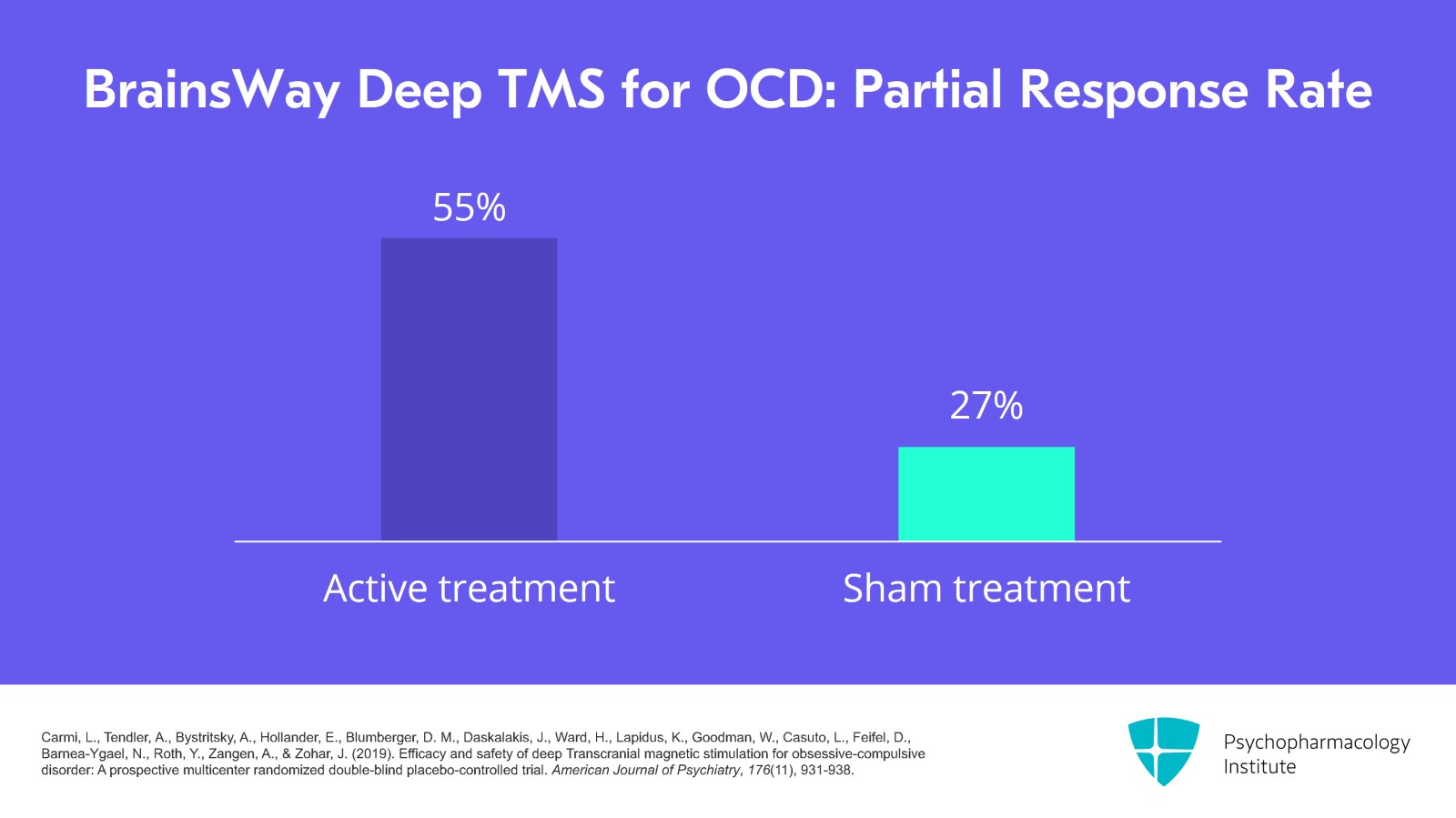
The partial response rate in the study was about 55% for active versus 27% for sham. Ten weeks, the improvement as measured by the Clinical Global Impression Scale was maintained.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
Slide 10 of 13
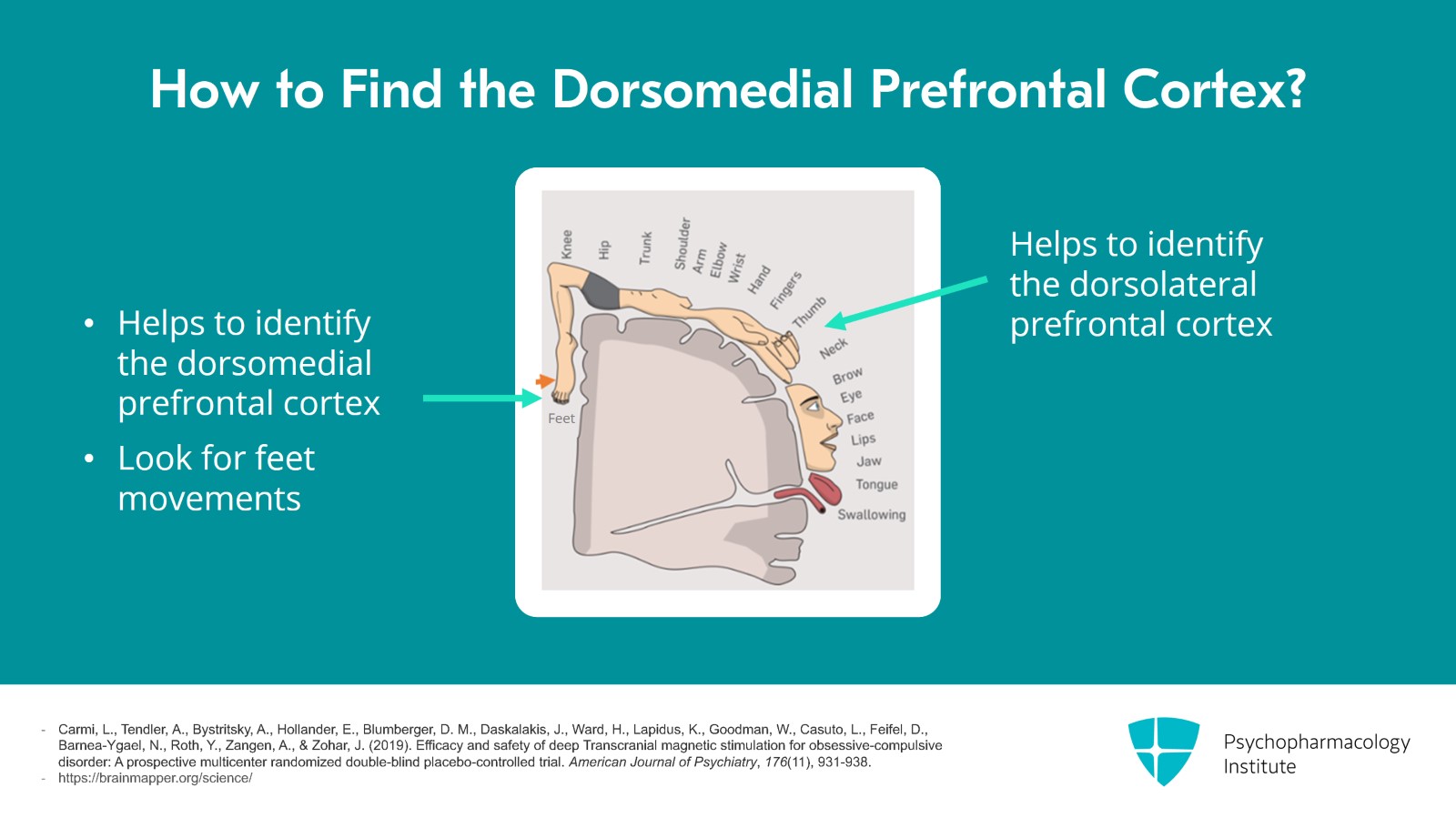
So how do we find the dorsomedial prefrontal cortex? So if you look at the homunculus, you'll see that the thumb identifies a location where we can move anteriorly 5.5 cm and that approximates the dorsolateral prefrontal cortex. Likewise, finding where we can get the feet to move helps us approximate the dorsomedial prefrontal cortex. So this position will be closer to midline. Motor threshold determination is the same as for depression except we look for feet movements.
References:
- Carmi, L., Tendler, A., Bystritsky, A., Hollander, E., Blumberger, D. M., Daskalakis, J., Ward, H., Lapidus, K., Goodman, W., Casuto, L., Feifel, D., Barnea-Ygael, N., Roth, Y., Zangen, A., & Zohar, J. (2019). Efficacy and safety of deep Transcranial magnetic stimulation for obsessive-compulsive disorder: A prospective multicenter randomized double-blind placebo-controlled trial. American Journal of Psychiatry, 176(11), 931-938.
- https://brainmapper.org/science/
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 11 of 13
Key points. So there are now three TMS devices FDA approved for adjunctive treatment of OCD. The treatment location is the dorsomedial prefrontal cortex.
Slide 12 of 13
Each treatment is preceded by provoking anxiety to a level of 4 to 7 out of 10; 10 is worst. Insurance companies might not be ready to cover TMS for OCD yet.
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.