Slides and Transcript
Slide 1 of 15
In the next section, I want to talk about how we adjust medications for opioid use disorder in patients with liver disease. And then, smoking cessation pharmacotherapy.
Slide 2 of 15
Buprenorphine is metabolized in the liver and plasma concentrations for both transmucosal buprenorphine and bup-naloxone, concentrations are increased and the half-lives are prolonged in moderate to severe hepatic impairment. But unless you’re seeing side effects, there’s no real safety concern with increased levels of buprenorphine.
References:
- Kumar, R., Viswanath, O., & Saadabadi, A. (2024, June 8). Buprenorphine. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK459126/
- Substance Abuse and Mental Health Services Administration. (n.d.). Medications for opioid use disorder: Quick start guide. U.S. Department of Health and Human Services. https://tinyurl.com/25h5dczt
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 3 of 15
Interestingly, the naloxone in the blood is affected to a greater degree than the buprenorphine. So the magnitude of the difference increases with the degree of hepatic impairment. Therefore, the risk of a precipitated withdrawal when you’re adding buprenorphine and naloxone to somebody with severe hepatic impairment or the risk of side effects favors the use of the mono-product especially for induction.
References:
- Kumar, R., Viswanath, O., & Saadabadi, A. (2024, June 8). Buprenorphine. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK459126/
- Substance Abuse and Mental Health Services Administration. (n.d.). Medications for opioid use disorder: Quick start guide. U.S. Department of Health and Human Services. https://tinyurl.com/25h5dczt
Slide 4 of 15
In severe hepatic impairment, it’s recommended we reduce the dose by half of what you normally use for buprenorphine or bup-nal and you double the titration increments, monitor for overdoses and toxicity.
References:
- Kumar, R., Viswanath, O., & Saadabadi, A. (2024, June 8). Buprenorphine. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK459126/
- Fareed, A., Vayalapalli, S., Casarella, J., & Drexler, K. (2012). Effect of buprenorphine dose on treatment outcome. Journal of Addictive Diseases, 31(1), 8–18. https://doi.org/10.1080/10550887.2011.642758
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 5 of 15
How about for transdermal buprenorphine? It’s not studied in severe hepatic impairment. Consider an alternative analgesic ’cause you can’t really adjust the dose very quickly. Now, transdermal, remember, is mainly used in pain management. Extended-release injections, it’s not recommended with moderate or severe hepatic impairment, again, because the formulation doesn’t allow for rapid dose adjustment. So if you overshoot and somebody becomes sedated, severely constipated, etc., you have a hard time having that abate.
References:
- Substance Abuse and Mental Health Services Administration. (n.d.). Medications for opioid use disorder: Quick start guide. U.S. Department of Health and Human Services. https://tinyurl.com/25h5dczt
Slide 6 of 15
With the development of moderate to severe hepatic impairment, monitor closely for these signs and symptoms of any overdose. If signs and symptoms occur within two weeks of administration, removal of the depo may be required. I’ve never tried that. I have to refer somebody to a surgeon.
References:
- Fareed, A., Vayalapalli, S., Casarella, J., & Drexler, K. (2012). Effect of buprenorphine dose on treatment outcome. Journal of Addictive Diseases, 31(1), 8–18. https://doi.org/10.1080/10550887.2011.642758
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 7 of 15
With the use of buprenorphine, elevations in liver function tests are rare and they’re not clinically significant. They may be more common in those who you’re treating with buprenorphine who have a comorbid hepatitis C virus infection. The manufacturer’s prescribing information does indicate that hepatitis with jaundice has been reported in those receiving buprenorphine including going on to all the way to hepatic failure. But they didn’t really control in these studies well for those that have comorbid IV drug use, the hepatitises B and C. And so I really don’t see liver problems with buprenorphine.
References:
- Fareed, A., Vayalapalli, S., Casarella, J., & Drexler, K. (2012). Effect of buprenorphine dose on treatment outcome. Journal of Addictive Diseases, 31(1), 8–18. https://doi.org/10.1080/10550887.2011.642758
Slide 8 of 15
Evaluate liver functions prior to the initiate of buprenorphine for opiate use disorder and periodically during treatment. I actually do this only every six months when somebody is on buprenorphine. The manufacturer recommends for the extended-release injectable buprenorphine monthly monitoring of liver function.
References:
- Substance Abuse and Mental Health Services Administration. (n.d.). Medications for opioid use disorder: Quick start guide. U.S. Department of Health and Human Services. https://tinyurl.com/25h5dczt
- Fareed, A., Vayalapalli, S., Casarella, J., & Drexler, K. (2012). Effect of buprenorphine dose on treatment outcome. Journal of Addictive Diseases, 31(1), 8–18. https://doi.org/10.1080/10550887.2011.642758
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 9 of 15
For methadone, while it’s hepatically metabolized, surprisingly, the pharmacokinetics have not been extensively evaluated in those with hepatic insufficiency. So the good rule of thumb since in severe hepatic dysfunction you have decreased clearance of methadone is to start low and go slow, monitor for signs of sedation and respiratory suppression.
References:
- Laks, J., & Alford, D. P. (2024). Treatment of opioid use disorder in patients with liver disease. Clinical Liver Disease, 23(1), e0207. https://doi.org/10.1097/CLD.0000000000000207
- U.S. Food and Drug Administration. (2016). Methadone hydrochloride tablets: Prescribing information. https://tinyurl.com/y2pktaae
Slide 10 of 15
In Child-Pugh class C, use very cautious initiation starting at half your standard dose. In those with liver transplantation, we shouldn’t deny people this valuable therapeutic option.
References:
- Laks, J., & Alford, D. P. (2024). Treatment of opioid use disorder in patients with liver disease. Clinical Liver Disease, 23(1), e0207. https://doi.org/10.1097/CLD.0000000000000207
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 11 of 15
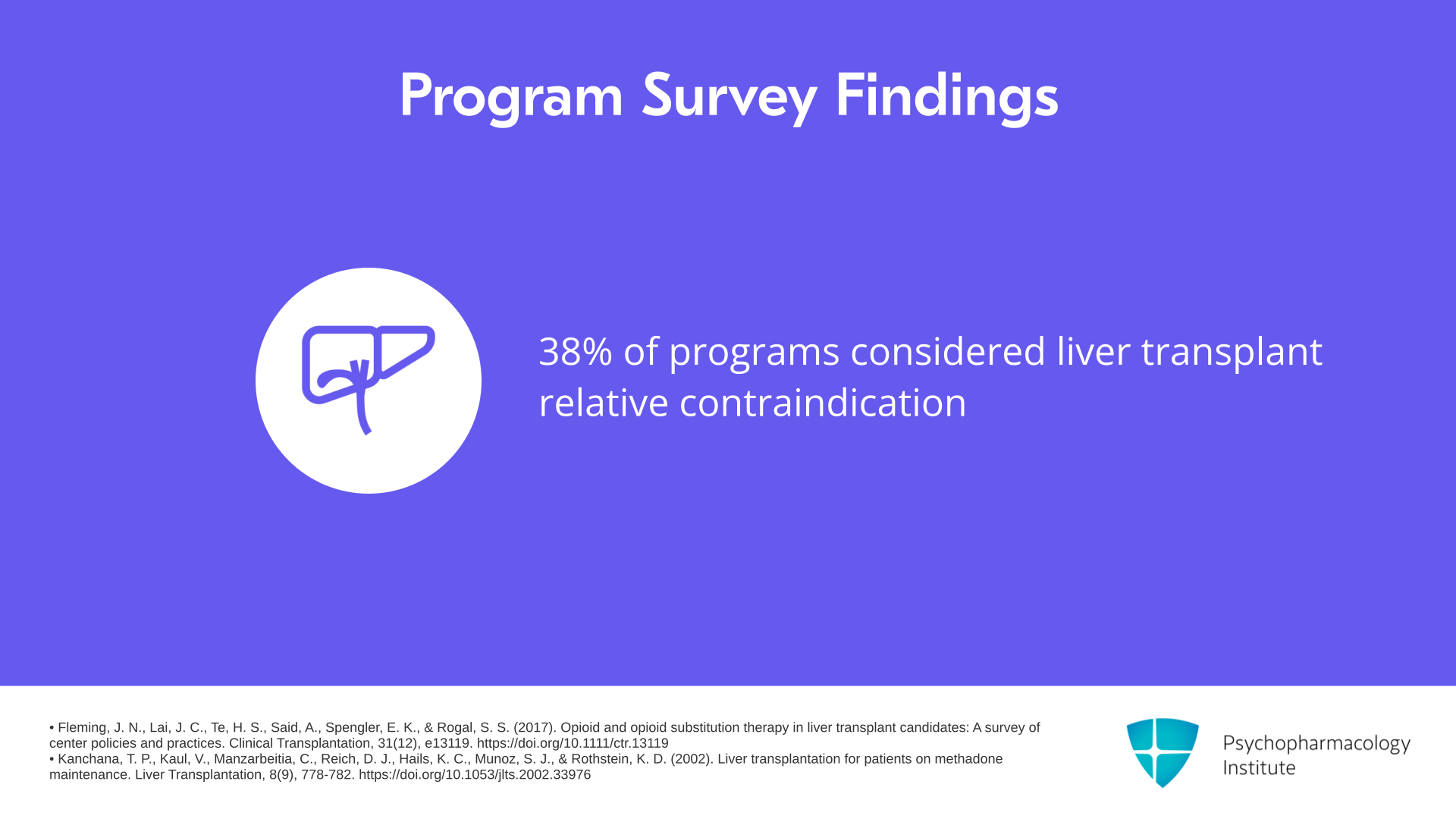
There’s a 2017 survey which is Fleming et al., 2017, 38% of programs considered a relative contraindication if somebody had had liver transplant for starting them on methadone. One said it was an absolute contraindication which just is not accurate based on the clinical information.
References:
- Fleming, J. N., Lai, J. C., Te, H. S., Said, A., Spengler, E. K., & Rogal, S. S. (2017). Opioid and opioid substitution therapy in liver transplant candidates: A survey of center policies and practices. Clinical Transplantation, 31(12), e13119. https://doi.org/10.1111/ctr.13119
- Kanchana, T. P., Kaul, V., Manzarbeitia, C., Reich, D. J., Hails, K. C., Munoz, S. J., & Rothstein, K. D. (2002). Liver transplantation for patients on methadone maintenance. Liver Transplantation, 8(9), 778-782. https://doi.org/10.1053/jlts.2002.33976
Slide 12 of 15
If you look at very small studies, 38 patients on methadone, only 2 on buprenorphine, showed good graft function when they were 18 months after transplant on these medications. But based on liver impairment, you still may have to reduce the MOUD doses. But if the liver function is restored by the transplantation, MOUD options are back on the table.
References:
- Laks, J., & Alford, D. P. (2024). Treatment of opioid use disorder in patients with liver disease. Clinical Liver Disease, 23(1), e0207. https://doi.org/10.1097/CLD.0000000000000207
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.
Slide 13 of 15
And then finally, I never want to forget smoking cessation pharmacotherapy. There’s no recommendation in the presence of liver disease to adjust the doses of either varenicline or nicotine replacement treatments – patch, gum, lozenge. But bupropion in Child-Pugh B, C, you should reduce the dose by about half. And with mild hepatic impairment, consider reducing the dose. It’s not recommended to use the highest dose of bupropion XL which is the 450 mg tab.
References:
- Huecker, M. R., Smiley, A., & Saadabadi, A. (2024, September 2). Bupropion. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK470212/
- Sprague, D., & Bambha, K. (2012). Drug-induced liver injury due to varenicline: A case report. BMC Gastroenterology, 12, Article 65. https://doi.org/10.1186/1471-230X-12-65
- Voican, C. S., Corruble, E., Naveau, S., & Perlemuter, G. (2014). Antidepressant-induced liver injury: a review for clinicians. The American Journal of Psychiatry, 171(4), 404-415. https://doi.org/10.1176/appi.ajp.2013.13050709
Slide 14 of 15
Remember, smoking more than alcohol use is the number one cause of physical mortality in those with substance use disorders in the world. In fact, in the world without substance use disorders, smoking is the number 2 cause. Many would say it’s the number 1 but they forget about obesity and sedentary lifestyle, which is the number 1 because of cardiovascular disease and cancer.
References:
- Peacock, A., Leung, J., Larney, S., Colledge, S., Hickman, M., Rehm, J., Giovino, G. A., West, R., Hall, W., Griffiths, P., Ali, R., Gowing, L., Marsden, J., Ferrari, A. J., Grebely, J., Farrell, M., & Degenhardt, L. (2018). Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction, 113(10), 1905-1926. https://doi.org/10.1111/add.14234
Free Files
Download PDF and other files
Success!
Check your inbox, we sent you all the materials there.