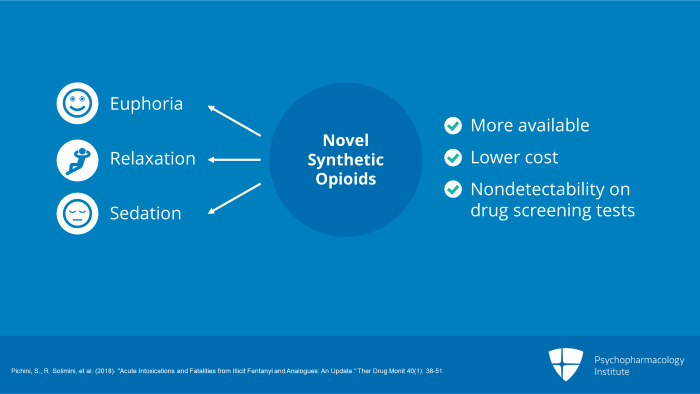
Now, let’s turn to section eight, Novel Synthetic Opioids: Their Clinical Manifestations, Diagnosis and Treatment. Novel synthetic opioids users seek the same subjective experience as do other opioid users that is euphoria or high, relaxation and sedation. Many opioid users prefer the experience of heroin or prescription opioid analgesics to that of fentanyl or its analogs but use synthetic opioids because of their greater availability especially with the increasing crackdown on prescription opioid analgesics. Other advantages are their lower cost in the illicit marketplace and non-detectability on drug screening tests.
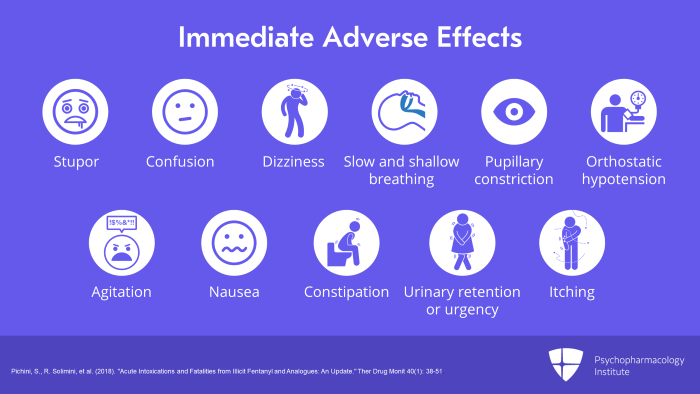
The immediate adverse effects are the same as those with other opiates such as stupor, confusion, dizziness, slow and shallow breathing, pupillary constriction or miosis, orthostatic hypotension, agitation, nausea, constipation, urinary retention or urgency and itching or pruritus.
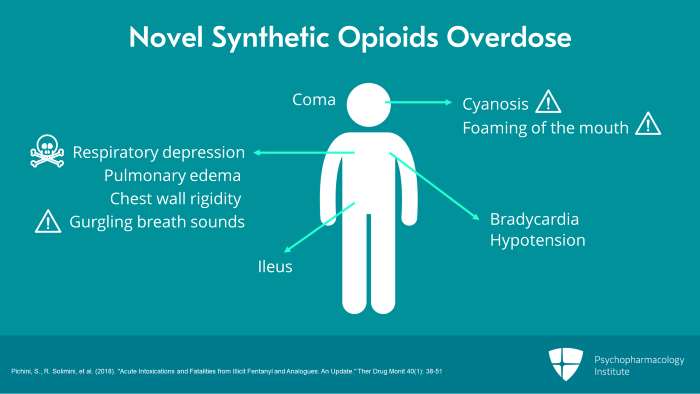
Overdose on novel synthetic opioids like overdose on other opioids is associated with respiratory depression, cyanosis, bradycardia and coma. Less common are hypotension, pulmonary edema and decreased intestinal motility or ileus. It is usually coma or respiratory depression that brings novel synthetic opioid users to medical attention with the latter resulting in death. High doses may also cause chest wall rigidity which can hinder resuscitation efforts. Cases of eventually fatal overdose may present with blue coloration of the lips from anoxia, gurgling breath sounds and foaming of the mouth.

Some cases of intoxication with novel synthetic opioids without the fentanyl chemical structure such as MT-45 have been reported with symptoms not usually associated with opioid intoxication such as dry eyes, dermatitis and elevated liver enzymes. However, given the small number of such cases and the uncertain purity of the drug, it is possible these adverse effects were not due to the synthetic opioid itself but were due to some other drug or contaminant.

Novel synthetic opioids are used by a variety of routes of administration so they may be sold in a variety of dosing formulations such as powder for intranasal or injection administration, tablet or capsule for oral administration, absorbed on blotter paper or on a patch for transdermal administration. Because of their high potency, a dose of synthetic opioid may occupy very little volume.


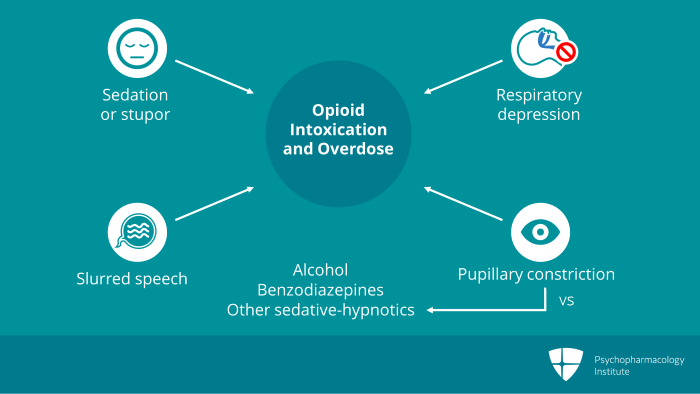
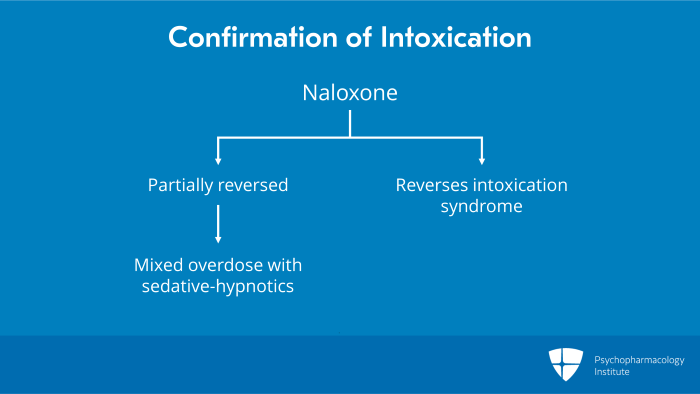

The specific treatment for novel synthetic opioid intoxication or overdose is naloxone, a selective mu opioid receptor antagonist which blocks the action of the opioid at the receptor thereby reversing the intoxication. The clinical goal is prompt reversal of life threatening manifestations such as respiratory depression and coma without precipitating severe acute opioid withdrawal.

Naloxone is currently available in four different dosage forms. First, there is a liquid solution containing 0.4 mg/mL. Second, there is a single dose autoinjector available in a 2.0 mg dose. Third, there is a multidose nasal atomizer which delivers a 1 mg in 1 mL dose and fourth, there is a single dose nasal spray available in 0.4 or 2.0 mg doses both contained in 0.1 mL.
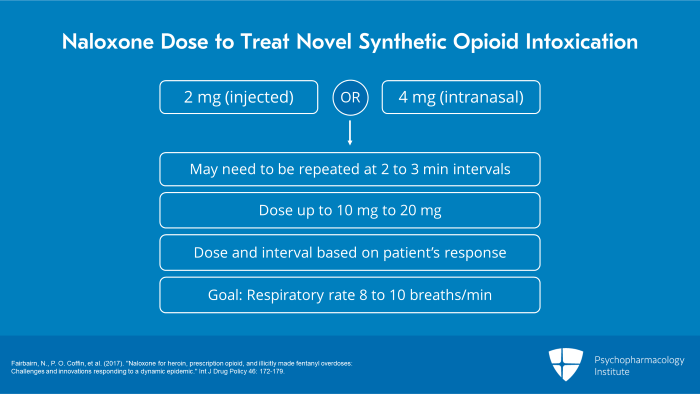
The optimal naloxone dose to treat novel synthetic opioid intoxication or overdose is not yet established. Because of the higher potency of novel synthetic opioids, experts recommend an initial dose of 2 mg injected or 4 mg intranasal rather than the 0.4 mg intravenous dose often recommended for heroin overdose. The naloxone dose may need to be repeated several times at two to three-minute intervals especially if the patient has taken novel synthetic opioids orally which have a longer duration of action. Some patients have needed cumulative naloxone doses of up to 10 to 20 mg. In every case, the dose and dosing interval should be titrated based on the patient’s actual response with the goal of restoring the respiratory rate to 8 to 10 breaths per minute.

Of key importance is administering naloxone as soon as possible as synthetic opioids may induce life threatening respiratory depression within minutes. The patient should be monitored for at least one to two hours after resuscitation with periodic checking of vital signs although not necessarily in a medical setting. This ensures prompt re-administration of naloxone and provision of oxygen and intravenous fluids should this become clinically indicated. Referral to evaluation for opioid use disorder and long-term treatment is essential as the strongest risk factor for fatal synthetic opioid overdose is a prior overdose.

Abrupt cessation of chronic or excessive synthetic opioid use often results in a withdrawal syndrome with features similar to but more intense than those associated with cessation of use of conventional opioids. The risk of withdrawal increases if the patient has been increasing their amount of use because of the development of tolerance. Acute withdrawal can also be precipitated by administration of mu opioid receptor antagonists such as naloxone.


…sweating, piloerection, commonly known as goosebumps, diarrhea, abdominal cramps, lacrimation or excessive tearing, rhinorrhea or nasal congestion, pupillary dilation or mydriasis and yawning. These are also the typical signs and symptoms of conventional opioid withdrawal. The presence of mydriasis, lacrimation and rhinorrhea distinguishes opioid withdrawal from sedative-hypnotic withdrawal.

We are not aware of any published clinical trials of treatment for novel synthetic opioid withdrawal but the treatments available for withdrawal from conventional opioids are likely to be helpful. Several clinical trials of the mu opioid receptor agonist, methadone or buprenorphine which are also used for the long-term treatment of opioid use disorder, show that short-term treatment is effective for opioid withdrawal including withdrawal from prescription semi-synthetic opioid analgesics.

Clinical management involves observation and monitoring until withdrawal symptoms have resolved and the patient is comfortable. In more severe cases, supportive and symptomatic treatment may also be needed such as intravenous fluids for dehydration and acetaminophen for pain or headache. Depression and suicidal ideation usually resolve as withdrawal wanes unless there is a comorbid mood disorder. Antidepressant treatment should be reserved for cases with depression persisting more than several days or a known independent mood disorder.


There is no such formal diagnosis in the DSM-5 or ICD-10 but the diagnosis could be made by generalizing from the criteria provided for opioid and other substance use disorders.
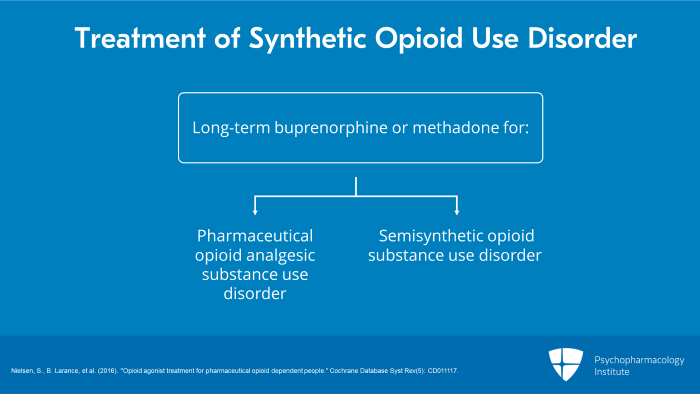
Several clinical trials show that long-term treatment with buprenorphine or methadone is effective for treatment of substance use disorder on pharmaceutical opioid analgesics including semi-synthetic opioids and is significantly more effective than psychological treatment alone.

Now, let us summarize the key points. First, overdose can cause life threatening coma and respiratory depression so it needs immediate treatment. Intoxication can be distinguished from other designer drug intoxication by pupillary constriction and prompt response to naloxone. Naloxone at an initial dose of 2 mg injected or 4 mg intranasal is the indicated treatment for synthetic opioid overdose. And finally, novel synthetic opioid withdrawal or use disorder may be treated with buprenorphine or methadone.
References:
- Pichini, S., R. Solimini, et al. (2018). “Acute Intoxications and Fatalities from Illicit Fentanyl and Analogues: An Update.” Ther Drug Monit 40(1): 38-51.
- Fairbairn, N., P. O. Coffin, et al. (2017). “Naloxone for heroin, prescription opioid, and illicitly made fentanyl overdoses: Challenges and innovations responding to a dynamic epidemic.” Int J Drug Policy 46: 172-179.
- Image
- Gowing, L., R. Ali, et al. (2017). “Buprenorphine for managing opioid withdrawal.” Cochrane Database Syst Rev 2: CD002025.
- Nielsen, S., B. Larance, et al. (2016). “Opioid agonist treatment for pharmaceutical opioid dependent people.” Cochrane Database Syst Rev(5): CD011117.



