Mirtazapine (Remeron) is a noradrenergic and specific serotonergic antidepressant (NaSSA), it doesn’t have effects as monoamine reuptake inhibitor. A significant feature is its effect as histamine 1 antagonist. This antihistamine effect is linked to sedation and weight gain.
Mirtazapine is commonly used in the elderly population. In this group of patients, insomnia and low weight might benefit from sedation and weight gain.
Mirtazapine has no significant drug-drug interactions, this makes it attractive for use in combination with other antidepressants as augmenting option.
Pharmacology and MOA
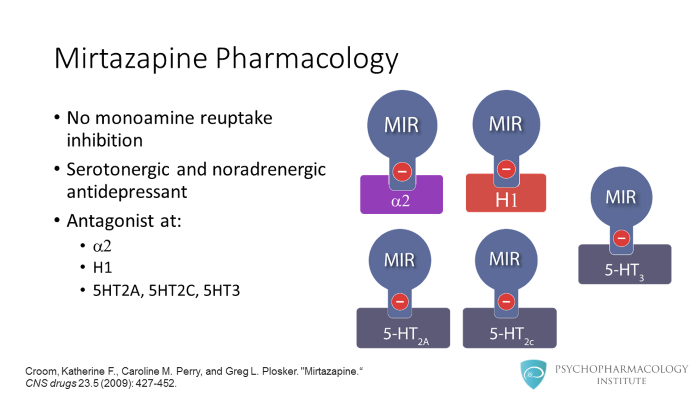
Mirtazapine’s pharmacology is different from SSRIs, SNRIs or tricyclics. What makes it substantially different is the fact that mirtazapine has no monoamine reuptake inhibition properties. However, it is a serotonergic and noradrenergic antidepressant, this is because mirtazapine blocks receptors that modulate release of these neurotransmitters. Regarding its binding profile, mirtazapine is mostly an antagonist. The receptors involved include: alpha 2, histamine 1, and the serotonin receptors 5HT2A, 5HT2C and 5HT3. In the next slides we’ll see how mirtazapine enhances serotonergic and noradrenergic activity through blockade of alpha 2 receptors.
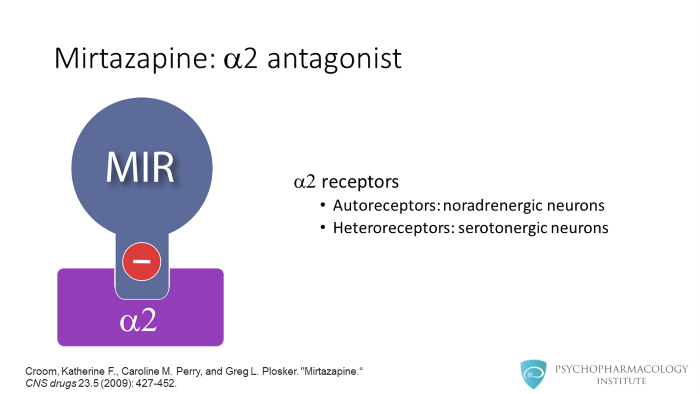
As I mentioned, mirtazapine is an alpha 2 antagonist. It blocks alpha 2 autoreceptors in noradrenergic neurons and alpha 2 heteroreceptors in serotonergic neurons.
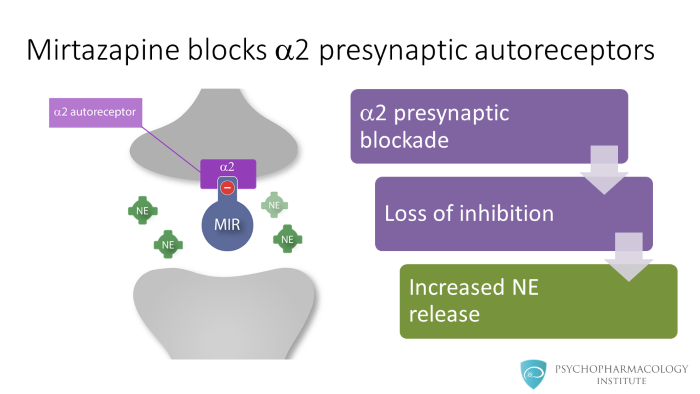
We can see in the image on the left how mirtazapine blocks alpha 2 presynaptic autoreceptors at a noradrenergic neuron. What’s the effect of this? Normally, activation of alpha 2 receptors triggers inhibitory signals that reduce norepinephrine release, if this signal is turned off, then the noradrenergic neuron loses its inhibition. This means that when mirtazapine blocks alpha 2 receptors it blocks the inhibitory signal, which increases norepinephrine release to the synaptic cleft.
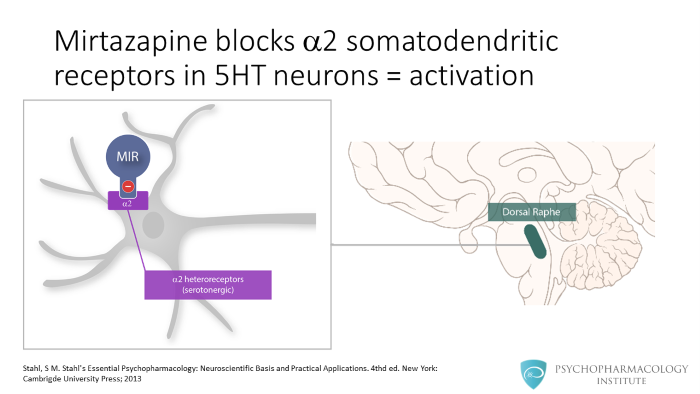
Let’s go now to alpha 2 heteroreceptors. Mirtazapine blocks alpha 2 somatodendritic receptors in serotonergic neurons. Serotonergic neurons are also inhibited by alpha 2 receptors, these are located in the somatodentric region. As the right picture shows, an important site of serotonergic cell bodies is the dorsal raphe. There are serotonergic projections from the dorsal raphe to the cortex. So, what is the effect of alpha 2 heteroreceptors blockade? Mirtazapine disinhibits serotonergic neurons in the dorsal raphe, leading to increased serotonin release in the cortex.
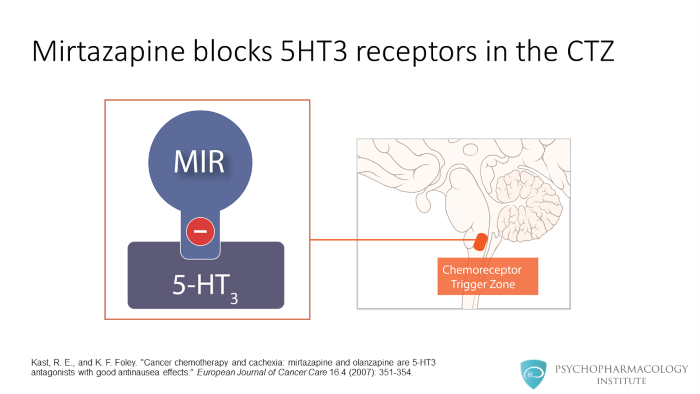
Mirtazapine blocks serotonin receptors, in this image the 5HT3 receptor is shown. Activation 5HT3 receptors at the chemoreceptor trigger zone is linked to vomiting. 5HT3 antagonism can be beneficial in cancer patients suffering from nausea and vomiting associated with chemotherapy.

Mirtazapine is also antagonist at 5HT2A and 5HT2C receptors. Blockade of 5HT2C receptors has been linked to antidepressant effects.
Clinical Uses
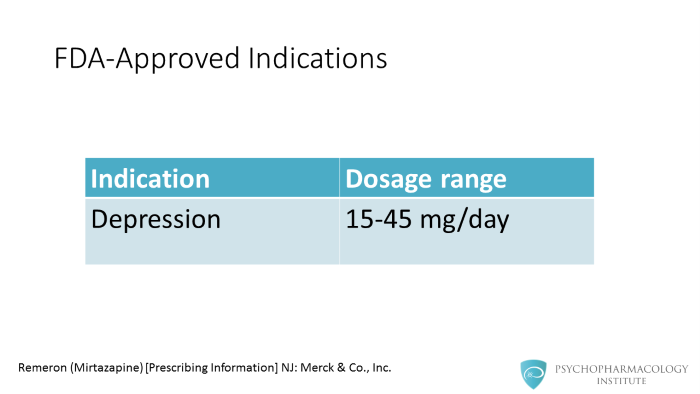
Mirtazapine was approved by the FDA in 1996 for major depressive disorder in a dosage range going from 15-45 mg/day. In some European countries mirtazapine maximum approved dose is 60 mg/day.

Mirtazapine is also used as augmenting agent. It is relatively easy to combine with other antidepressants, the venlafaxine + mirtazapine combination has been called “California Rocket Fuel” . Since it has no activity on monoamine reuptake it has a complementary mechanism of action with SSRIs and SNRIs. So what’s the evidence supporting its use as augmenting agent? The STAR*D trial showed that the mirtazapine + venlafaxine combination outperformed tranylcypromine. However, this finding was not statistically significant. Case series of 32 patients studied the venlafaxine mirtazapine combination, measuring response rate based on the CGI scale. 44% of patients responded at 4 weeks, while 50% responded at 8 weeks. So, as a conclusion even though the combination of mirtazapine with venlafaxine makes pharmacological sense, the evidence is limited to case reports. Its use can be considered according to clinical judgment of the prescriber.

Mirtazapine is approved only for depression. There are studies supporting its use in anxiety disorders such as panic disorder, generalized anxiety disorder and social anxiety disorder. Small size randomized controlled trials support the use of mirtazapine as add-on to antipsychotics for the treatment of negative symptoms in schizophrenia. The mechanism of action of antiemetic drugs involves 5HT3 antagonism. Mirtazapine is an interesting option for depressed patients suffering from chemotherapy induced nausea and vomiting. Because of its histamine 1 antagonist properties mirtazapine is sometimes used for the treatment of insomnia. We should keep in mind that mirtazapine can be useful in depressed patients with insomnia, but shouldn’t be used as primary treatment for insomnia without depression.
Pharmacokinetics

Mirtazapine has a half-life of 20 to 40 hours. It has no active metabolites and doesn’t inhibit significantly any CYP 450 isoenzyme. It is metabolized by three CYP 450 isoenzyme 1A2, 2D6 and 3A4. It has no significant drug-drug interactions, which makes it attractive for its use in combination with other antidepressants.
Adverse Effects

As we saw in the mechanism of action section, mirtazapine is a histamine 1 antagonist. Antihistaminic effects are among mirtazapine’s most prominent features. The blockade of histamine 1 receptors is linked to sedation, increased appetite, weight gain and dry mouth. • Sedation: this effect can appear in up to 50% of patients during the first of treatment. • Increased appetite and weight gain: this can occur in 15-25% of patients. Tricyclic antidepressants,
, and mirtazapine are strongly associated with increased risk of weight gain. There were reports in premarketing studies of three cases of agranulocytosis, this is reflected in a prescribing information warning. An association has not been yet established.

Mirtazapine has some advantages in terms of its side effects profile. When compared to SSRIs or SNRIs, mirtazapine is associated with significantly lower frequency of antidepressant-induced sexual dysfunction, gastrointestinal disturbances and insomnia.
Prescribing information
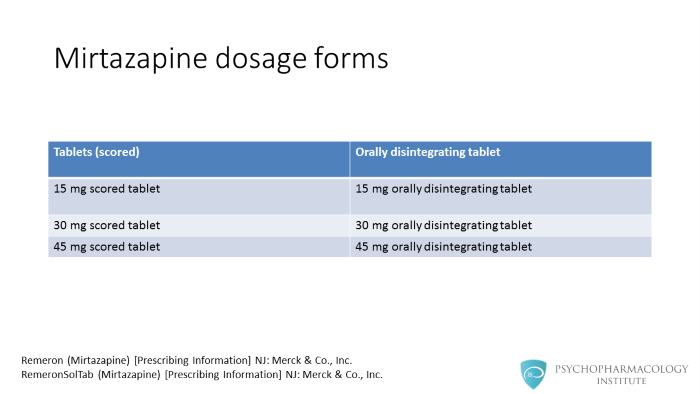
Mirtazapine is available scored tablets and orally disintegrating tablets. Scored tablets of 15, 30 and 45 mg. Orally disintegrating tablets of 15, 30 and 45 mg.
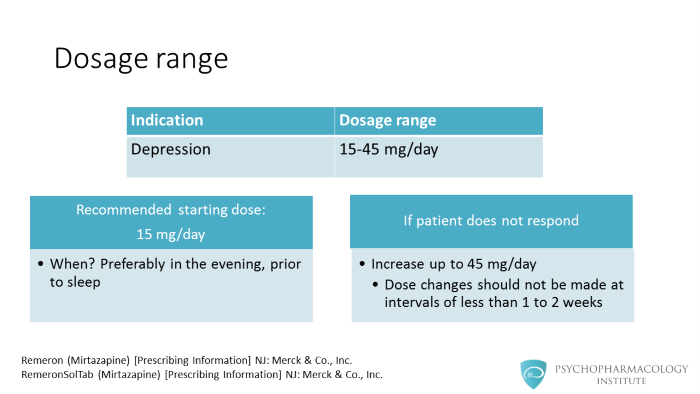
Mirtazapine’s dosage range goes from 15 to 45 mg/day. The recommended starting dose is of 15 mg/day . Because of mirtazapine’s sedative properties it is given preferably in the evening, prior to sleep. If the patient doesn’t respond, the manufacturer recommends dose increases up to a maximum of 45 mg/day. These dose changes should not be made at intervals of less than 1 to 2 weeks.

The key points for this presentation are the following: • Mirtazapine’s mechanism of action involves blockade of alpha 2, 5HT2A, 5HT2C and 5HT3 receptors. • Mirtazapine is FDA-approved only for depression, but there is evidence of efficacy for anxiety disorders and negative symptoms of schizophrenia. • It can be used as augmenting agent in combination with venlafaxine. • Prominent side effects include sedation and weight gain. • It is dosed between 15-45 mg/day. This tutorial is also available in spanish:
Mirtazapina: mecanismo de acción, indicaciones, efectos adversos, farmacocinética y posología
References
- Croom, Katherine F., Caroline M. Perry, and Greg L. Plosker. “Mirtazapine.“ CNS drugs 23.5 (2009): 427-452.
- Stahl, S M. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications . 4thd ed. New York: Cambrigde University Press; 2013
- Kast, R. E., and K. F. Foley. “Cancer chemotherapy and cachexia: mirtazapine and olanzapine are 5‐HT3 antagonists with good antinausea effects.” European Journal of Cancer Care 16.4 (2007): 351-354.
- Remeron (Mirtazapine) [Prescribing Information] NJ: Merck & Co., Inc.
- McGrath, Patrick, et al. “Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR* D report.” American Journal of Psychiatry 163.9 (2006): 1531-1541.
- Hannan, Noel, et al. “Venlafaxine—mirtazapine combination in the treatment of persistent depressive illness.” Journal of Psychopharmacology 21.2 (2007): 161-164.
- Marin, H; Escobar, J. Clinical Psychopharmacology : A Practical Approach . 1st edition. World Scientific Publishing Company, 2013
- Phan, S. V., & Kreys, T. J. M. (2011). Adjunct mirtazapine for negative symptoms of schizophrenia . Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 31(10), 1017-1030.
- Thase,M “Mirtazapine”, in Kaplan and Sadock’s Comprehensive Textbook of Psychiatry. 9th ed. Philadelphia: Lippincott Williams & Wilkins, 2009.
- RemeronSolTab (Mirtazapine) [Prescribing Information] NJ: Merck & Co., Inc.



